Advertisements
Advertisements
प्रश्न
How do enzymes catalyse a chemical reaction in the living system? Explain drug target interaction taking the example of enzyme as target.
उत्तर
In the catalytic activity, enzymes perform the following two major functions:
(i) The first function of an enzyme is to hold the substrate molecule for a chemical reaction. the active sites of the enzymes hold the substrate molecule in a suitable position so that it can be attacked by the reagent effectively.
The substrate molecules bind to the amino acid residues of the protein present the active site of the enzyme through a variety of interactions such as hydrogen bonding, dipole-dipole interactions, van der Waals' interactions, and ionic bonding.
These binding forces should be strong enough to hold the substrate long enough so that the enzyme can catalyze the reaction, but weak enough to allow the products to depart after their formation.
(ii) The second function of the enzyme Is to provide functional groups which win attack me substrate to carry out the chemical reactions. This function is carried out by some other amino acid residues of protein present on the active site of the enzyme. These provide free amino groups to attack substrate and bring about the chemical reaction. If the amino acid serine is present nearby the substrate held on the active site, then its — OH group is free to act as a nucleophile in the enzyme-catalyzed reaction.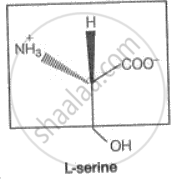
The part of the amino acid which lies outside the box act as a nucleophile in enzyme-catalyzed reactions, but the part of the amino acid which is enclosed in the box is involved in the formation of a peptide bond in a protein molecule.
Drugs can inhibit these activities by blocking site of the enzyme and prevent the binding of substrate. Such drugs are called enzyme inhibitors.
APPEARS IN
संबंधित प्रश्न
Name the macromolecules that are chosen as drug targets.
Which forces are involved in holding the drugs to the active site of enzymes?
Which site of an enzyme is called allosteric site?
Why are certain drugs called enzyme inhibitors?
Explain the role of allosteric site in enzyme inhibition?
What happens when the bond formed between an enzyme and an inhibitor is a strong covalent bond?
Assertion: Enzymes have active sites that hold substrate molecule for a chemical reaction.
Reason: Drugs compete with natural substrate by attaching covalently to the active site of enzyme.
Assertion: Competitive inhibitors compete with natural substrate for their attachment on the active sites of enzymes.
Reason: In competitive inhibition, inhibitor binds to the allosteric site of the enzyme.
Assertion: Non-competitive inhibitor inhibits the catalyic activity of enzyme by binding with its active site.
Reason: Non-competitive inhibitor changes the shape of the active site in such a way that substrate can’t recognise it.
What are enzyme inhibitors? Classify them on the basis of their mode of attachments on the active site of enzymes. With the help of diagrams explain how do inhibitors inhibit the enzymatic activity.
