Advertisements
Advertisements
प्रश्न
How is ethyne prepared in the laboratory?
उत्तर
Ethyne Is prepared by the reaction of calcium carbide with water
\[\ce{CaC2 + 2H2O -> C2H2 + Ca(OH)2}\]
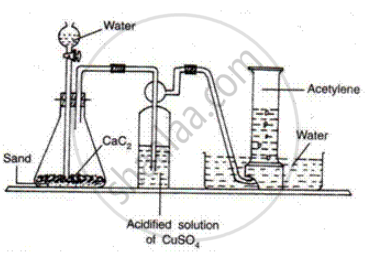
APPEARS IN
संबंधित प्रश्न
The molecular formula of acetic acid is _________ .
(a) CH3COOH
(b) CH3 – CH3
(c) C6H6
(d) C2H4
A student takes about 6 ml of distilled water in each of the four test tubes P, Q, R and S. He then dissolves an equal amount of four different salts namely, sodium chloride in 'P', potassium chloride in 'Q', calcium chloride in 'R' and magnesium chloride in 'S'. Next, he then adds 10 drops of soap solution to each test tube and shakes its contents. The test tubes in which scum (insoluble substance) is formed with soap are:
(A) P and Q
(B) Q and R
(C) R and S
(D) Q and S
Complete the following chemical equations:
C2H5OH+CH3COOH`("conc."H_2SO_4)/`>
Draw the structure of butanoic acid.
Three organic compounds A, B and C have the following molecular formulae: C4H10O
Which compound contains a carboxyl group? Write its name and structural formula.
If you take a pinch of sodium hydrogen carbonate powder in a test-tube and add drop-by-drop acetic acid to it, what would you observe immediately? List any two main observations.
Acetic acid smells like:
(1) a banana
(2) vinegar
(3) an orange
(4) a lemon
Name the first four members of alphatic carboxylic acid.
What type of compound is formed by the reaction between acetic acid and an alcohol?
Convert ethane to acetic acid.
