Advertisements
Advertisements
Question
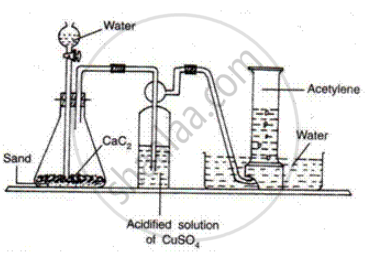
How is ethyne prepared in the laboratory?
Solution
Ethyne Is prepared by the reaction of calcium carbide with water
\[\ce{CaC2 + 2H2O -> C2H2 + Ca(OH)2}\]
APPEARS IN
RELATED QUESTIONS
Which of the following will give brisk effervescence with sodium hydrogen carbonate and why?
CH3COOH, CH3CH2OH
Write the IUPAC name of the following:
\[\begin{array}{cc}
\ce{CH3}\\
|\\
\ce{CH3 -C -CH3}\\
|\\
\ce{CH3}
\end{array}\]
Write the names of three compounds which can be oxidised directly or in stages to produce acetic acid.
Give two tests to show that CH3COOH is acidic in nature ?
Give the structural formula of Acetic acid.
Write a balanced chemical equation for the following:
Monochloroethane is hydrolysed with aqueous KOH.
Write the molecular formula of the given compound.
Sodium ethanoate
Write the molecular formula of the given compound.
Ethanoic acid
Which of the following substance produces brisk effervescence with baking soda solution?
Write the chemical equation for the ethanol to ethanoic acid of an oxidation reaction.
