Advertisements
Advertisements
Question
Which of the following will give brisk effervescence with sodium hydrogen carbonate and why?
CH3COOH, CH3CH2OH
Solution
CH3COOH (ethanoic acid) gives brisk effervescence with sodium hydrogencarbonate. It reacts with sodium hydrogencarbonate to give sodium ethanoate, carbon dioxide and water. Effervescence is due to carbon dioxide evolved.
APPEARS IN
RELATED QUESTIONS
Write three different chemical reactions showing the conversion of ethanoic acid to sodium ethanoate. Write balanced chemical equation in each case. Write the name of the reactants and the products other ethanoic acid and sodium ethanoate in each case.
What are esters ?
What is the common name of methanol?
Name the products formed and give appropriate chemical equations for the following:
sodium reacting with ethyl alcohol.
Write the name of the first three members of the carboxylic acid series.
Name the compound formed when acetic acid and ethanol react together?
What is the action of acetic acid with litmus paper?
Explain the following reaction with an example.
Saponification
The reaction between acetic acid and sodium carbonate is shown in the following figure.
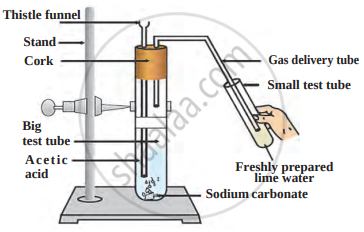
Answer the questions with the help of a diagram.
- Which gas does come out as effervescence in the big test tube?
- What is the colour change in the lime water present in the small test tube?
- Write the related reaction.
How is ethanol to ethanoic acid an oxidation reaction different from the reaction in which ethanol burns in the presence of oxygen?
