Advertisements
Advertisements
Question
Write the molecular formula of the given compound.
Ethanoic acid
Solution
Ethanoic acid- CH3COOH or C2H4O2
APPEARS IN
RELATED QUESTIONS
Complete the following chemical equations:
C2H5OH+CH3COOH`("conc."H_2SO_4)/`>
Complete the following chemical equations: C2H5OH+O2→
Complete the following chemical equations: CH3COOH+NaHCO3→
Complete the following equation:
`CH_3 COOH + CH_2 H_5 OH` 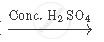
Which of the following molecular formula corresponds to ethylbutanoate ester?
(a) C5H10O2
(b) C6H12O2
(c) C7H14O2
(d) C8H16O2
Consider the following organic compounds:
HCHO, C2H5OH, C2H6, CH3COOH, C2H5CI
Choose two compounds which can react in the presence of conc. H2SO4 to form an ester. Give the name and formula of the ester formed.
What do you observe when acetic acid is added to ethyl alcohol in the presence of sulphuric acid?
Acetic acid solution turns:
(1) blue litmus red
(2) red litmus blue
(3) blue litmus colourless
(4) red litmus colourless
Explain the following term with example.
Monomer
Choose the correct answer:
Select the acid which contains four hydrozen atoms in it.
State how the following conversions can be carried out:
Ethene to Ethyl alcohol
Give reason for the following :
Ethyne is more reactive than ethane
Choose the correct alternative and rewrite the following:
Some acetic acid is treated with solid NaHCO3, the resulting solution will be _________________.
Ester is formed by the reaction between ______.
Write an equation showing saponification.
A spatula full of sodium carbonate is taken in a test tube and 2 mL of dilute ethanoic acid is added to it.
Write a chemical equation for the reaction.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Give the balanced chemical equation of the reaction.
Oxidation of ethanol by acidified potassium dichromate.
