Advertisements
Advertisements
प्रश्न
How is propene converted into 1- bromopropane and 2 - bromopropane?
उत्तर
The addition of hydrogen halide to an unsymmetrical alkene gives two products.
Propene on reaction with hydrogen bromide forms 80% 2-bromopropane (isopropyl bromide) and 20% 1-bromopropane (n-propyl bromide).

APPEARS IN
संबंधित प्रश्न
The preparation of alkyl fluoride from alkyl chloride, in presence of metallic fluorides is known as ______________.
How do you convert: Propene to 1-iodopropane
Write the structure of the major product in each of the following reaction :
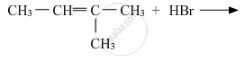
Draw the structure of major monohalo product in each of the following reactions :
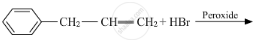
Write a short note on Sandmeyer’s reaction.
Rectified spirit is a mixture of ____________.
Gem-dibromide is ____________.
Conant Finkelstein reaction for the preparation of alkyl iodide is based upon the fact that:
The boiling points of alcohols are higher than those of hydrocarbons of comparable masses due to ______.
Which of the following is halogen exchange reaction?
Which of the following are secondary bromides?
(i) \[\ce{(CH3)2 CHBr}\]
(ii) \[\ce{(CH3)3C CH2Br}\]
(iii) \[\ce{CH3CH(Br)CH2CH3}\]
(iv) \[\ce{(CH3)2 CBrCH2CH3}\]
Alkyl halides are prepared from alcohol by treating with ______.
(i) HCl + ZnCl2
(ii) Red P + Br
(iii) H2SO4 + KI
(iv) All the above
Aryl chlorides and bromides can be easily prepared by electrophilic substitution of arenes with chlorine and bromine respectively in the presence of Lewis acid catalysts. But why does preparation of aryl iodides requires presence of an oxidising agent?
Identify the products A and B formed in the following reaction:
\[\ce{CH3 - CH2 - CH = CH - CH3 + HCl -> A + B}\]
Predict the major product formed when HCl is added to isobutylene. Explain the mechanism involved.
Match the structures given in Column I with the names in Column II.
| Column I | Column II | |
| (i) | 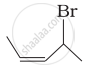 |
(a) 4-Bromopent-2-ene |
| (ii) | 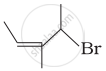 |
(b) 4-Bromo-3-methylpent-2-ene |
| (iii) | 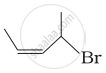 |
(c) 1-Bromo-2-methylbut-2-ene |
| (iv) | 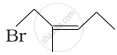 |
(d) 1-Bromo-2-methylpent-2-ene |
Assertion: Phosphorus chlorides (tri and penta) are preferred over thionyl chloride for the preparation of alkyl chlorides from alcohols.
Reason: Phosphorus chlorides give pure alkyl halides.
Which is gem-dihalide?
The alkyl halide which does not give white precipitate with alcoholic AgNO3 solution is :-
Benzoyl chloride is is prepared from benzoic acid by
Name the possible alkenes which will yield 1-chloro-1-methylcyclohexane on their reaction with HCl. Write the reactions involved.
Predict the reagent for carrying out the following transformations:
Ethanoic acid to 2-chloroethanoic acid
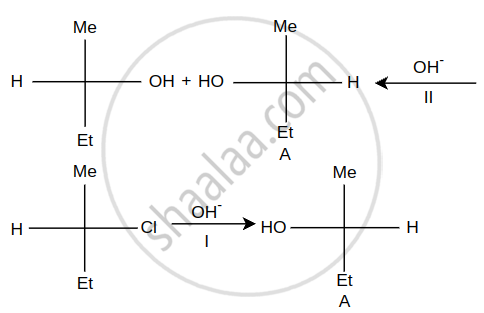
Steps I and II are ______.
In Finkelstein Reaction, which reactants are used?
\[\ce{C2H5Cl + AgF -> C2H5F + AgCl}\] The above reaction is called ______.
The major product of the following reaction is:
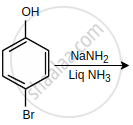
Arrange the following in increasing order of reactivity towards nitration
- p-xylene
- bromobenzene
- mesitylene
- nitrobenzene
- benzene
Choose the correct answer from the options given below:
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
