Advertisements
Advertisements
Question
How is propene converted into 1- bromopropane and 2 - bromopropane?
Solution
The addition of hydrogen halide to an unsymmetrical alkene gives two products.
Propene on reaction with hydrogen bromide forms 80% 2-bromopropane (isopropyl bromide) and 20% 1-bromopropane (n-propyl bromide).

APPEARS IN
RELATED QUESTIONS
The preparation of alkyl fluoride from alkyl chloride, in presence of metallic fluorides is known as ______________.
What is the action of bromine in alkaline medium on
i. CH3CH2NO2
ii. 
How do you convert the following: Prop-1-ene to 1-fluoropropane
In the following reaction, the compound used in the reaction for synthesizing ethyl fluoride is:
______ \[\ce{+ AgF -> H3C - F + AgBr}\]
Rectified spirit is a mixture of ____________.
The solution of a chemical compound reacts with AgNO3 solution to form a white precipitate of Y which dissolves in NH4OH to give a complex Z. When Z is treated with dilute HNO3, Y reappears. The chemical compound X can be:
The catalyst used in the preparation of an alkyl chloride by the action of dry HCl on alcohol is ____________.
Alkyl halides are prepared from alcohol by treating with ______.
(i) HCl + ZnCl2
(ii) Red P + Br
(iii) H2SO4 + KI
(iv) All the above
Aryl chlorides and bromides can be easily prepared by electrophilic substitution of arenes with chlorine and bromine respectively in the presence of Lewis acid catalysts. But why does preparation of aryl iodides requires presence of an oxidising agent?
Identify the products A and B formed in the following reaction:
\[\ce{CH3 - CH2 - CH = CH - CH3 + HCl -> A + B}\]
Which of the following haloalkanes reacts with aqueous \[\ce{KOH}\] most easily? Explain giving reason.
Which of the following compounds would undergo SN1 reaction faster and why?
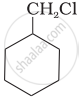 |
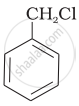 |
| (A) | (B) |
tert-Butylbromide reacts with aq. \[\ce{NaOH}\] by SN1 mechanism while n-butylbromide reacts by SN2 mechanism. Why?
Assertion: Phosphorus chlorides (tri and penta) are preferred over thionyl chloride for the preparation of alkyl chlorides from alcohols.
Reason: Phosphorus chlorides give pure alkyl halides.
Which of the following is the most stable free radical?
Which compound would undergo dehydrohalogenation with strong base to give the alkene shown below as the only alkene product?
CH3 – CH2CH = CH – CH3
The alky halide is converted into an alcohol by
The alkyl halide which does not give white precipitate with alcoholic AgNO3 solution is :-
The product of reaction of alcoholic silver nitrite with ethyl bromide are:
Benzoyl chloride is is prepared from benzoic acid by
Name the possible alkenes which will yield 1-chloro-1-methylcyclohexane on their reaction with HCl. Write the reactions involved.
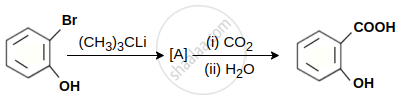
In the given conversion the compound A is:
The major product of the following reaction is:
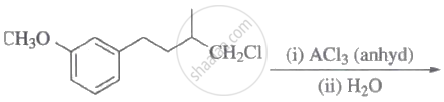
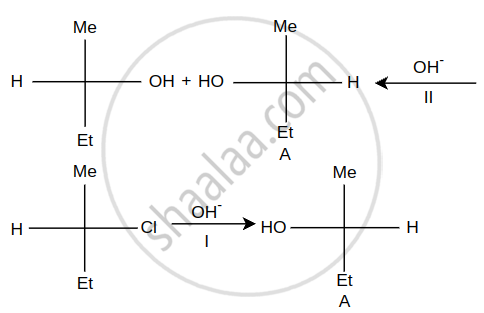
Steps I and II are ______.
In Finkelstein Reaction, which reactants are used?
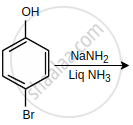
The major product of the following reaction is:
Arrange the following in increasing order of reactivity towards nitration
- p-xylene
- bromobenzene
- mesitylene
- nitrobenzene
- benzene
Choose the correct answer from the options given below:
