Advertisements
Advertisements
प्रश्न
How will you convert sodium acetate to methane?
उत्तर
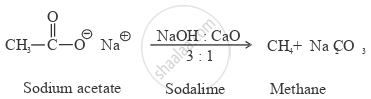
APPEARS IN
संबंधित प्रश्न
Arrange the following in the increasing order of their reactivity towards nucleophilic addition reaction:
C6H5COCH3, CH3-CHO, CH3COCH3
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Benzaldehyde, p-Tolualdehyde, p-Nitrobenzaldehyde, Acetophenone.
Hint: Consider steric effect and electronic effect.
What is meant by the following term? Give an example of the reaction in the following case.
Cyanohydrin
What is meant by the following term? Give an example of the reaction in the following case.
Ketal
What is meant by the following term? Give an example of the reaction in the following case.
Imine
What is meant by the following term? Give an example of the reaction in the following case.
Schiff’s base
How will you bring about the following conversion in not more than two steps?
Bromobenzene to 1-Phenylethanol
Give plausible explanation for the following:
Cyclohexanone forms cyanohydrin in good yield but 2, 2, 6 trimethylcyclohexanone does not.
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Explain the mechanism of alkaline hydrolysis of tert-butyl bromide with energy profile diagram.
How are the following compounds prepared?
acetophenone from benzene
How are the following compounds prepared?
benzaldehyde from benzoyl chloride
Write balanced chemical equations for action of ammonia on - acetaldehyde
Write balanced chemical equations for action of ammonia on - acetone
Alkenes 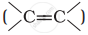 and carbonyl compounds
and carbonyl compounds 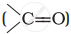 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
Identify the compounds A, B and C in the following reaction.
\[\ce{CH3 - Br ->[Mg/ether] (A) ->[(i) CO][(ii) Water] (B) ->[CH3OH/H+][Δ] (C)}\]
The pH of blood does not appreciably change by a small addition of acid or base because
Which of the following has the most acidic hydrogen?
What happens when ethanal is treated with excess ethanol and acid?
The product of following reaction is
\[\ce{CH3 - CH = CH - CH2 - CHO ->[i) LiAlH4][ii) H3O+]}\] ______?
Out of p-tolualdehyde and p-nitrobenzaldehyde, which one is more reactive towards nucleophilic addition reactions, why?
Write the structure of the product formed when acetone reacts with 2, 4 DNP reagent.
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structures of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
