Advertisements
Advertisements
प्रश्न
How will you distinguish between the following pairs of terms: Tetrahedral and octahedral voids
उत्तर
Tetrahedral voids and octahedral voids:
| Tetrahedral voids | Octahedral voids |
|
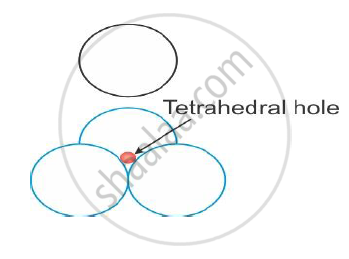
The tetrahedral void is the name given to the vacant space formed by touching of four spheres, as shown in the figure.
|
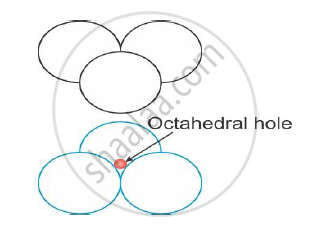
Octahedral voids are formed by combination of voids of two triangular layers of atoms, when one triangular layer is placed above another, as shown in the figure,
|
APPEARS IN
संबंधित प्रश्न
If the radius of the octachedral void is r and radius of the atoms in close packing is R, derive relation between r and R.
Total number of voids in 0.5 mole of a compound which forms hexagonal close packed structure is ____________.
The Ca2+ and F– are located in CaF2 crystal, respectively at face centered cubic lattice points and in ____________.
In which of the following crystals alternate tetrahedral voids are occupied?
In the hexagonal close-packed structure of a metallic lattice, the number of nearest neighbours of a metallic atom is ____________.
If Germanium crystallises in the same way as diamond, then which of the following statement is not correct?
In the cubic close packing, the unit cell has ______.
In which of the following arrangements octahedral voids are formed?
(i) hcp
(ii) bcc
(iii) simple cubic
(iv) fcc
Which of the following is not true about the voids formed in 3 dimensional hexagonal close packed structure?
(i) A tetrahedral void is formed when a sphere of the second layer is present above triangular void in the first layer.
(ii) All the triangular voids are not covered by the spheres of the second layer.
(iii) Tetrahedral voids are formed when the triangular voids in the second layer lie above the triangular voids in the first layer and the triangular shapes of these voids do not overlap.
(iv) Octahedral voids are formed when the triangular voids in the second layer exactly overlap with similar voids in the first layer.
Show that in a cubic close packed structure, eight tetrahedral voids are present per unit cell.