Advertisements
Advertisements
प्रश्न
How will you represent first order reactions graphically.
उत्तर
i. The differential rate law for the first-order reaction A → P is 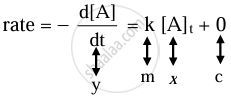
The equation is of the form y = mx + c. A plot of rate versus [A]t is a straight line passing through the origin. The slope of straight line = k.
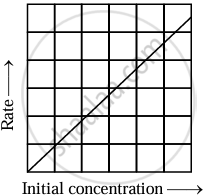 |
| Variation of rate with [A] |
ii. The integrated rate law is
k = `2.303/t log_10 ["A"]_0/["A"]_"t"`
On rearrangement, the equation becomes
`(kt)/2.303 = log_10 ["A"]_0 - log_10 ["A"]_"t"`
Hence,
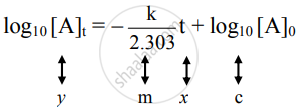
The equation is of the straight line. A graph of `log_10[A]_t` versus t yields a straight line with slope `-"k"/2.303` and y-axis intercepts as log10[A]0.
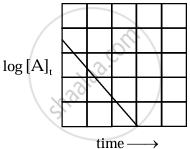 |
| Variation of `log_10 [A]_t` with time |
iii. Rearranging the integrated rate law equation, we get
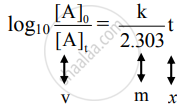
The equation has a straight-line form y = mx. Hence, the graph of `log_10 ([A]_0)/([A]_t)` versus t is a straight line passing through the origin.
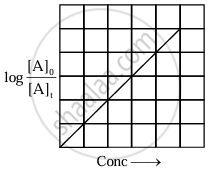 |
| Variation of `log_10 ([A]_0)/([A]_t)` with time |
APPEARS IN
संबंधित प्रश्न
Answer the following in brief.
How will you represent the zeroth-order reaction graphically?
Answer the following in brief.
What are the units of the rate constant?
For first order reaction, the rate constant for the decomposition of N2O5 is 6 × 10–4 s –1. The half-life period for decomposition in seconds is ______.
Give one example of pseudo first-order reaction.
What is half life of first order reaction if time required to decrease concentration of reactants from 0.8 M to 0.2 M is 12 hours?
Explain with the help of a potential energy diagram that the catalyst increases the rate of the reaction.
For a first-order reaction \[\ce{A -> B}\] the rate constant is x min−1. If the initial concentration of A is 0.01 M, the concentration of A after one hour is given by the expression.
The decomposition of phosphine (PH3) on tungsten at low pressure is a first-order reaction. It is because the
The rate constant of a reaction is 5.8 × 10−2 s−1. The order of the reaction is ____________.
A zero order reaction is 20% complete in 20 minutes. Calculate the value of the rate constant. In what time will the reaction be 80% complete?
The integrated rate law is a direct relationship between ____________ and ____________.
What is the value of rate constant of first order reaction, if it takes 15 minutes for consumption of 20% of reactants?
In a first order reaction, the concentration of the reactant, decreases from 0.8 mol dm−3 to 0.4 mol dm−3 in 15 minutes. The time taken for the concentration to change from 0.1 mol dm−3 to 0.025 mol dm−3 is ____________.
The rate and the rate constants of the reaction, \[\ce{A + 2B -> C + D}\] are 8 × 10−3 mol dm−3 s−1 and 2 × 10−3 s−1 respectively. The concentrations of A and B are 4 mol dm−3 each. The overall order of the reaction is ____________.
For a zero order reaction, the plot of [A]t vs t is linear. The slope of the line is equal to ____________.
The rate of formation of B at time t for reaction \[\ce{2A -> 3B}\] is equal to ____________.
Which among the following is an example of zero order reaction?
Which among the following is an example of pseudo first order reaction?
In a reaction \[\ce{N2_{(g)} + 3H2_{(g)} -> 2NH3_{(g)}}\], if the rate of disappearance of N2(g) is 2.6 × 10−4 M/s, the rate of disappearance of H2(g) in M/s is ____________.
For the first order reaction A → B, the rate constant is 0.25 s-1, if the concentration of A is reduced to half, the value of rate constant will be ________.
A certain zero order reaction has rate constant 0.025 M s-1. What will be the concentration of reactant 'A' after 15 seconds, if initial concentration is 0.50 M?
What is the unit of rate constant for the zero order reaction?
Rate constant for zero order reaction is 2 × 10-2 mol L-1 s-1. If the concentration of the reactant after 25 sec. is 0.5 M, what is the initial concentration of reactant?
The integrated rate law is a direct relationship between time and ______
Which of the following reactions is not of the first order?
Which among the following reactions is an example of pseudo first order reaction?
Consider the following reaction.
\[\ce{SO2(g) + 1/2 O2(g) <=>[K1] SO3(g)}\]
\[\ce{2SO3(g)<=>[K2] 2SO2(g) + O2(g)}\]
What is the relation between K1 and K2?
Which of the following correctly represents integrated rate law equation for a first order reaction in a gas phase?
