Advertisements
Advertisements
प्रश्न
How would you separate gold from a mixture of gold and copper filling?
उत्तर
Gold will dissolve only in aqua regia i.e., a mixture of conc. hydrochloric acid and conc. nitric acid in 1:3 but copper will dissolve in nitric acid.
To separate gold from a mixture of gold and platinum add some nitric acid to the mixture, gold will remain undissolved hence can be filtered.
APPEARS IN
संबंधित प्रश्न
Give a balanced chemical equation for Action of conc. Nitric acid on Sulphur
Fill in the blanks using the appropriate words given below :
(Sulphur dioxide, nitrogen dioxide, Nitric oxide, Sulphuric acid)
Hot, concentrated nitric acid reacts with sulphur to form ______________.
State one relevant observation for the following:
When crystals of copper nitrate are heated in a test tube.
What is : aqua regia
- Write a balanced chemical equation for the laboratory preparation of nitric acid.
- In the preparation of nitric acid from \[\ce{KNO3}\], concentrated hydrochloric acid is not used in place of concentrated sulphuric acid. Explain why?
- Conc. nitric acid prepared in laboratory is yellow in colour. Why? How is this colour removed?
- Give reasons for the following:
In the laboratory preparation of nitric acid, the mixture of concentrated sulphuric acid and sodium nitrate should not be heated very strongly above 200°C.
Name the Following:
Product obtained by the catalytic oxidation of ammonia.
Write the equation to show the reaction between the following:
Between copper and concentrated nitric acid.
Write equation to show the reaction between the following:
Copper oxide and dilute nitric acid.
Write a balanced equation for following :
Action of cold and dilute nitric acid on copper
The diagram given below is a representation of the Industrial preparation of Nitric acid by Ostwald’s process. With respect to the process answer the following questions:
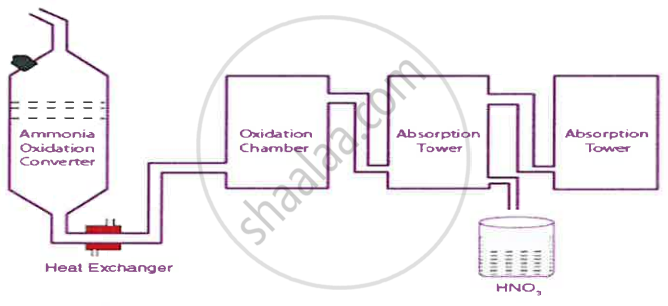
- Write the temperature and the catalyst required during the catalytic oxidation of ammonia.
- Give balanced chemical equation for the reaction occurring duringthe conversion of nitrogen dioxide to nitric acid.
