Advertisements
Advertisements
प्रश्न
Hydrogen is a good reducing agent: What do you understand by the above statement? Explain with the help of copper oxide as an example.
उत्तर
Hydrogen acts as a good reducing agent means, when hydrogen gas is passed over hot metallic oxides of copper, lead, iron, etc. it removes oxygen from them and thus reduces them to their corresponding metal.
Let us consider the following example, in each of which metallic oxide react with hydrogen. Metallic oxide act as oxidising agents and hydrogen acts as a reducing agents.
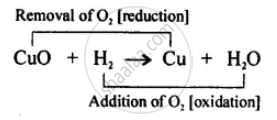
APPEARS IN
संबंधित प्रश्न
Complete and balance the following equation:
H2+____________→ 2HCI
FILL IN THE BLANK
Ammonia is used to make ................
Write the balanced equation and give your observation when the following metal reacts:
Magnesium with boiling water
Choose the correct option:
The composition of the nucleus of deuterium is
Using a burning candle and a jar of hydrogen – how would you prove experimentally that Hydrogen is a combustible gas.
Give balanced equation for the following conversion:
Iron [III] oxide to iron – using hydrogen.
Match the statements in List I with the appropriate answer in List II.
| List I | List II |
| 1. An atom of hydrogen | A: Chromic oxide |
| 2. A strong oxidising agent | B: Ammonia |
| 3. A promoter used in Bosch process | C: Iron |
| 4. A chemical used in the manufavture of fertilizers | D: One electron |
| 5. The catalyst used in production of a basic gas from nitrogen. | E: Dilute nitric acid |
When steam is passed over red-hot iron, magnetic oxide of iron and hydrogen are obtained. “The reaction between steam and red-hot iron is a Reversible Reaction.” What is meant by this statement.
Name a gas which burns in air or oxygen forming water.
Explain the following:
Two jars of H2 are collected – “one burns quietly and the other does not”.
