Advertisements
Advertisements
प्रश्न
If the total energy of radiation of frequency 1014 Hz is 6.63 J, Calculate the number of photons in the radiation.
उत्तर
Given:
E = 6.63 J, ν = 1014 Hz,
We know, h = 6.63 × 10–34 Js.
To find: Number of photons (n)
Formula: n = `"E"/"hv"`
Calculation:
Using formula,
n = `6.63/(6.63 xx 10^-34 xx 10^14)`
∴ n = 1020
The number of photons emitted in the radiation is 1020.
संबंधित प्रश्न
Is it always possible to see the photoelectric effect with a red light?
Given the following data for incident wavelength and the stopping potential obtained from an experiment on the photoelectric effect, estimate the value of Planck's constant and the work function of the cathode material. What is the threshold frequency and corresponding wavelength? What is the most likely metal used for emitter?
| Incident wavelength (in Å) | 2536 | 3650 |
| Stopping potential (in V) |
1.95 | 0.5 |
The maximum kinetic energy of the photoelectrons depends only on ______
Find the energy of photon which have momentum 2 × 10-16 gm-cm/sec.
Draw a neat labelled diagram of a schematic of the experimental setup for the photoelectric effect.
State Einstein’s photoelectric equation. Explain all characteristics of the photoelectric effect, on the basis of Einstein’s photoelectric equation.
The maximum velocity of photoelectron emitted is 4.8 m/s. If the e/m ratio of the electron is 1.76 × 1011 C/kg, then stopping potential is given by ______
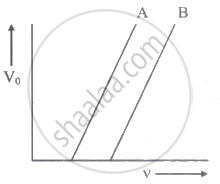
The following graph shows the stopping potential V0 versus frequency v for photoelectric emission from two metals A and B. The slope of each of the lines gives ______
If the maximum kinetic energy of emitted electrons in photoelectric effect is 3.2 × 10-19 J and the work-function for metal is 6.63 × 10-19 J, then stopping potential and threshold wavelength respectively are
[Planck's constant, h = 6.63 × 1034 J-s]
[Velocity of light, c = 3 × 108 `"m"/"s"`]
[Charge on electron= 1.6 × 10-19 C]
For photoelectric emission from certain metal, the cut-off frequency is v. If radiation of frequency 2v impinges on the metal plate, the maximum possible velocity of the emitted electron will be (m is the electron mass) ____________.
When a light of wavelength 4000 Å falls on a photoelectric emitter, photoelectrons are liberated. For another emitter, light of wavelength 6000 Å is sufficient for photo emission. The work functions of the two emitters are in the ratio of ____________.
A metal surface is illuminated by light of given intensity and frequency to cause photoemission. If the intensity of illumination is reduced to one-fourth of its original value then the maximum KE of the emitted photoelectrons would be ______.
The work function of a metal is 1.6 x 10-19 J. When the metal surface is illuminated by the light of wavelength 6400 Å, then the maximum kinetic energy of emitted photo-electrons will be (Planck's constant h = 6.4 x 10-34 Js) ____________.
A metal surface is illuminated by photons of energy 5 eV and 2.5 eV respectively. The ratio of their wavelengths is ____________.
When light of wavelength 'λ' is incident on a photosensitive surface, the stopping potential is 'V'. When light of wavelength '3λ' is incident on the same surface, the stopping potential is `"V"/6`. Threshold wavelength for the surface is _______.
The work function of a photosensitive material is 4.0 eV. The longest wavelength of light that can cause photon emission from the substance is (approximately) ____________.
The radiations of energies 1 eV and 2.5 eV are incident on a metal surface having work function 0.5 eV. The ratio of the maximum velocities of the emitted photo-electrons is ____________.
An electromagnetic wave of wavelength '`lambda`' is incident on a photosensitive surface of negligible work function. If the photoelectrons emitted from this surface have the de-Broglie wavelength '`lambda_1`' then ____________.
A light of frequency 'v' is incident on the metal surface whose threshold frequency is 'v0'. If v = v0, then [c = speed of light in medium] ____________.
In experiment of photoelectric effect, the stopping potential for incident yellow light of wavelength 5890 Å is 4 volt. If the yellow light is replaced by blue light of wavelength 4000 Å, the stopping potential is ____________.
In a photoelectric experiment, ultraviolet light of wavelength 280 nm is used with a lithium cathode having work function Φ = 2.5 eV. If the wavelength of incident light is switched to 400 nm, find out the change in the stopping potential.
(h = 6.63 × 10-34 Js, c = 3 × 108 ms-1)
In a photocell, frequency of incident radiation is increased by keeping other factors constant (v > v0), the stopping potential ______.
On a photosensitive material when frequency of incident radiation is increased by 30%, kinetic energy of emitted photoelectrons increases from 0.4 eV. The work function of the surface is ______.
Light of wavelength λ, which is less than threshold wavelength is incident on a photosensitive material. If incident wavelength is decreased so that emitted photoelectrons are moving with same velocity, then stopping potential will ______.
Light of two different frequencies whose photons have energies 1.3 eV and 2.8 eV respectively, successfully illuminate a metallic surface whose work function is 0.8 eV. The ratio of maximum speeds of emitted electrons will be ______.
The threshold frequency for a certain metal for photoelectric effect is 1.7 x 1015 Hz. When a light of frequency 2.2 x 1015 Hz is incident on the metal surface, the kinetic energy of the emitted photoelectrons is 3.3 x.10-19 J. Calculate Planck's constant.
Explain the experimental set-up of photoelectric effect.
In a photoelectric experiment, the stopping potential is 1.5V. What is the maximum kinetic energy of a photoelectron?
