Advertisements
Advertisements
प्रश्न
If the total energy of radiation of frequency 1014 Hz is 6.63 J, Calculate the number of photons in the radiation.
उत्तर
Given:
E = 6.63 J, ν = 1014 Hz,
We know, h = 6.63 × 10–34 Js.
To find: Number of photons (n)
Formula: n = `"E"/"hv"`
Calculation:
Using formula,
n = `6.63/(6.63 xx 10^-34 xx 10^14)`
∴ n = 1020
The number of photons emitted in the radiation is 1020.
संबंधित प्रश्न
If the frequency of incident light falling on a photosensitive material is doubled, then the kinetic energy of the emitted photoelectron will be ______.
What is the photoelectric effect?
Is it always possible to see the photoelectric effect with a red light?
Using the values of work function given in the following table, tell which metal will require the highest frequency of incident radiation to generate photocurrent.
Typical values of work function for some common metals
| Metal | Work function (in eV) |
| Potassium | 2.3 |
| Sodium | 2.4 |
| Calcium | 2.9 |
| Zinc | 3.6 |
| Silver | 4.3 |
| Aluminium | 4.3 |
| Tungsten | 4.5 |
| Copper | 4.7 |
| Nickel | 5.0 |
| Gold | 5.1 |
Photocurrent recorded in the microammeter in an experimental setup of the photoelectric effect vanishes when the retarding potential is more than 0.8 V if the wavelength of incident radiation is 4950 Å. If the source of incident radiation is changed, the stopping potential turns out to be 1.2 V. Find the work function of the cathode material and the wavelength of the second source.
Radiation of wavelength 4500 Å is incident on a metal having work function 2.0 eV. Due to the presence of a magnetic field B, the most energetic photoelectrons emitted in a direction perpendicular to the field move along a circular path of radius 20 cm. What is the value of the magnetic field B?
The maximum kinetic energy of the photoelectrons depends only on ______
Find the kinetic energy of the emitted electron, if in photoelectric effect energy of incident Photon is 4 eV and work function is 2.4 eV.
The maximum velocity of photoelectron emitted is 4.8 m/s. If the e/m ratio of the electron is 1.76 × 1011 C/kg, then stopping potential is given by ______
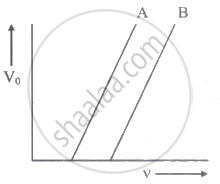
The following graph shows the stopping potential V0 versus frequency v for photoelectric emission from two metals A and B. The slope of each of the lines gives ______
The threshold frequency for a certain photosensitive metal is v0. When it is illuminated by light of frequency v = 2v0, the maximum velocity of photoelectrons is v0. What will be the maximum velocity of the photoelectrons when the same metal is illuminated by light of frequency
v = 5v0?
In photoelectric effect, for a light of different intensities but of same frequency, the stopping potential for a given metal is ____________.
The photo electric effect to take place for a metal, the minimum frequency required is 5.792 × 1014 Hz. A light of wavelength 6000 Å is incident on that metal surface. What is the corresponding frequency of light and will there be photoelectric emissions? [velocity of light = 3 × 108 m/s]
Photoelectrons are emitted from a photosensitive surface for the light of wavelengths λ1 = 360 nm and λ2 = 600 nm. What is the ratio of work functions for lights of wavelength 'λ1' to 'λ2'?
The work function of a substance is 4.0 eV. The longest wavelength of light that can cause photo-emission from this substance is approximately (h = 6.63 × 10-34 Js)[1eV = 1.6 × 10-19 J]
Following graphs show the variation of stopping potential corresponding to the frequency of incident radiation (F) for a given metal. The correct variation is shown in graph (v0 = Threshold frequency).
The lowest frequency of light that will cause the emission of photoelectrons from the surface of a metal (for which work function is 1.65 eV) will be ____________.
The work function of a photosensitive material is 4.0 eV. The longest wavelength of light that can cause photon emission from the substance is (approximately) ____________.
When a certain metallic surface is illuminated with monochromatic light of wavelength '`lambda`', the stopping potential for photoelectric effect is '3V0'. If the same surface is illuminated with a light of wavelength '`2 lambda`', the stopping potential is found as 'V0'. The threshold wavelength for this surface is ____________.
Is it always necessary to use red light to get a photoelectric effect?
Photoelectrons are observed to just emit out of a material surface when the light of 620 nm falls on it with the intensity of 100 W m-2. If the light of wavelength 400 nm is incident on the same material with an intensity of 1 W m-2, what would be the minimum reverse potential needed to stop the outflow of the electrons?
When ultraviolet light of wavelength 100 nm is incident upon a sample of silver metal, a potential difference of 7.7 volt is required to stop the photoelectrons from reaching the collector plate. The potential required to stop photo electrons when light of wavelength 200 nm is incident upon silver is ______.
Two radiations of photons energies 1 eV and 2.5 eV, successively illuminate a photosensitive metallic surface of work function 0.5 eV. The ratio of the maximum speeds of the emitted electrons is ______.
If the maximum kinetic energy of emitted electrons in the photoelectric effect is 2eV, the stopping potential will be ______.
Give Einstein's explanation of the photoelectric effect.
Explain the formation of clouds at high altitude.
