Advertisements
Advertisements
प्रश्न
Match the detergents given in Column I with their uses given in Column II.
| Column I | Column II |
(i) 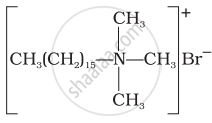 |
(a) Dishwashing powder |
(ii) 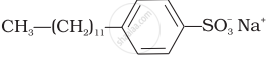 |
(b) Laundry soap |
| (iii) \[\ce{C17H33CO\overset{-}{O}\overset{+}{N}a + Na2CO3 + Rosin}\] | (c) Hair conditioners |
| (iv) \[\ce{CH3(CH2)16COO(CH2CH2O)nCH2CH2OH}\] | (d) Toothpaste |
उत्तर
| Column I | Column II |
(i)  |
(c) Hair conditioners |
(ii)  |
(d) Toothpaste |
| (iii) \[\ce{C17H33CO\overset{-}{O}\overset{+}{N}a + Na2CO3 + Rosin}\] | (b) Laundry soap |
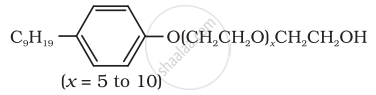
| (iv) \[\ce{CH3(CH2)16COO(CH2CH2O)nCH2CH2OH}\] | (a) Dishwashing powder |
Explanation:
(i) Hair shampoos/conditioners are made up of cationic detergents. These are quaternary ammonium salts of amines with chlorides, bromides or acetates, e.g., cetyltrimethyl ammonium bromide.
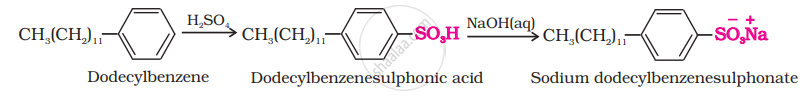
(ii) Anionic detergents are used in toothpaste e.g., sodium dodecylbenzene sulphonate. It can be prepared as follows.
\[\ce{\underset{Lauryl alcohol}{CH3(CH2)10CH2OH} ->[H2SO4] \underset{Laury hydrogensulphate}{CH3(CH2)10CH2OSO3H} ->[NaOH(aq)] \underset{(Anionic detergent)}{\underset{Sodium laurylsulphate}{CH3(CH2)10CH2OS\overset{-}{O}\overset{+}{N}a}}}\]
(iii) Laundry soaps contain fillers like sodium rosinate. Sodium silicate, borax and sodium carbonate. Sodium rosinate makes the soap to lather well.
(iv) Dishwashing powder are non-ionic detergents.
APPEARS IN
संबंधित प्रश्न
Why do soaps not work in hard water?
Explain the mechanism of cleansing action of soaps.
How soap is prepared?
Which of the following enhances leathering property of soap?
What is a soft soap?
How are transparent soaps manufactured?
Assertion: Transparent soaps are made by dissolving soaps in ethanol.
Reason: Ethanol makes things invisible.
Assertion: Sodium chloride is added to precipitate soap after saponification.
Reason: Hydrolysis of esters of long-chain fatty acids by alkali produces soap in colloidal form.
Green chemistry in day-to-day life is in the use of ______.
Self-cleansing windows are example of the ______.
