Advertisements
Advertisements
प्रश्न
Match the detergents given in Column I with their uses given in Column II.
| Column I | Column II |
(i) 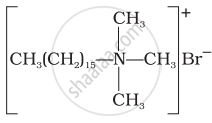 |
(a) Dishwashing powder |
(ii) 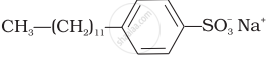 |
(b) Laundry soap |
| (iii) \[\ce{C17H33CO\overset{-}{O}\overset{+}{N}a + Na2CO3 + Rosin}\] | (c) Hair conditioners |
| (iv) \[\ce{CH3(CH2)16COO(CH2CH2O)nCH2CH2OH}\] | (d) Toothpaste |
उत्तर
| Column I | Column II |
(i)  |
(c) Hair conditioners |
(ii)  |
(d) Toothpaste |
| (iii) \[\ce{C17H33CO\overset{-}{O}\overset{+}{N}a + Na2CO3 + Rosin}\] | (b) Laundry soap |
| (iv) \[\ce{CH3(CH2)16COO(CH2CH2O)nCH2CH2OH}\] | (a) Dishwashing powder |
Explanation:
(i) Hair shampoos/conditioners are made up of cationic detergents. These are quaternary ammonium salts of amines with chlorides, bromides or acetates, e.g., cetyltrimethyl ammonium bromide.
(ii) Anionic detergents are used in toothpaste e.g., sodium dodecylbenzene sulphonate. It can be prepared as follows.
\[\ce{\underset{Lauryl alcohol}{CH3(CH2)10CH2OH} ->[H2SO4] \underset{Laury hydrogensulphate}{CH3(CH2)10CH2OSO3H} ->[NaOH(aq)] \underset{(Anionic detergent)}{\underset{Sodium laurylsulphate}{CH3(CH2)10CH2OS\overset{-}{O}\overset{+}{N}a}}}\]
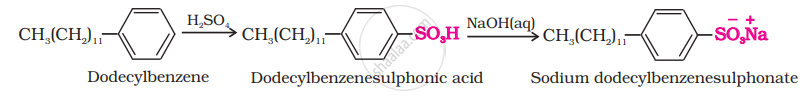
(iii) Laundry soaps contain fillers like sodium rosinate. Sodium silicate, borax and sodium carbonate. Sodium rosinate makes the soap to lather well.
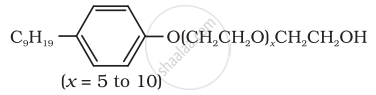
(iv) Dishwashing powder are non-ionic detergents.
APPEARS IN
संबंधित प्रश्न
Why is bithional added to soap?
Write the chemical equation for preparing sodium soap from glyceryl oleate . Structural formulae of the compounds are given below.
(C17H32COO)3C3H5 – Glyceryl oleate
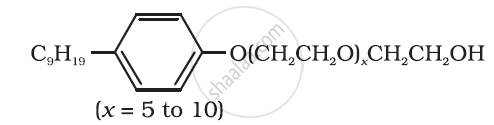
Following type of nom-ionic detergents are present in liquid detergents, emulsifying agents and wetting agents. Label the hydrophilic and hydrophobic parts in the molecule. Identify the functional group (s) present in the molecule.
Can you use soaps and synthetic detergents to check the hardness of water?
Which of the following enhances leathering property of soap?
Glycerol is added to soap. It functions ______.
Match the soaps given in Column I with items given in Column II.
| Column I | Column II |
| (i) Soap chips | (a) dried miniature soap bubbles |
| (ii) Soap granules | (b) small broken pieces of soap formed from melted soaps |
| (iii) Soap powder | (c) soap powder + abrasives + builders \[\ce{(Na2CO3,Na3PO4)}\] |
| (iv) Scouring soap | (d) soap powder + builders like \[\ce{Na2CO3}\] and \[\ce{Na3PO4}\] |
Assertion: Sodium chloride is added to precipitate soap after saponification.
Reason: Hydrolysis of esters of long-chain fatty acids by alkali produces soap in colloidal form.
Which of the following is not a correct statement?
Self-cleansing windows are example of the ______.
