Advertisements
Advertisements
प्रश्न
Match the reactions given in Column I with the suitable reagents given in Column II.
| Column I (Reactions) |
Column II (Reagents) |
| (i) Benzophenone Diphenylmethane | (a) \[\ce{LiAlH4}\] |
| (ii) Benzaldehyde 1-Phenylethanol | (b) \[\ce{DIBAL-H}\] |
| (iii) Cyclohexanone Cyclohexanol | (c) \[\ce{Zn(Hg)/Conc. HCl}\] |
| (iv) Phenyl benzoate Benzaldehyde | (d) \[\ce{CH3MgBr}\] |
उत्तर
| Column I (Reactions) |
Column II (Reagents) |
| (i) Benzophenone Diphenylmethane | (c) \[\ce{Zn(Hg)/Conc. HCl}\] |
| (ii) Benzaldehyde 1-Phenylethanol | (d) \[\ce{CH3MgBr}\] |
| (iii) Cyclohexanone Cyclohexanol | (a) \[\ce{LiAlH4}\] |
| (iv) Phenyl benzoate Benzaldehyde | (b) \[\ce{DIBAL-H}\] |
Explanation:
(i)
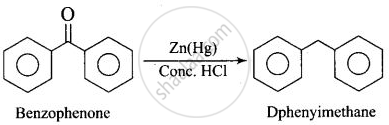
(ii)
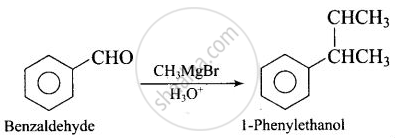
(iii)

(iv)
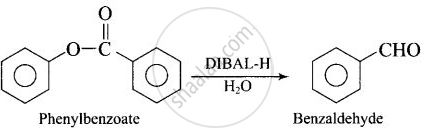
APPEARS IN
संबंधित प्रश्न
Write the structures of A and B in the following reactions
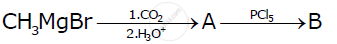
Show how the following compound can be converted to benzoic acid.
Acetophenone
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
Methyl benzoate
How is methoxy benzene prepared from carbolic acid?
What is the action of following reagents on glucose?
1. Bromine water
2. Hydroxylamine
Which is the most suitable reagent for the following conversion?
\[\begin{array}{cc}
\phantom{....................}\ce{O}\phantom{.....................................}\ce{O}\phantom{.}\\
\phantom{....................}||\phantom{......................................}||\phantom{.}\\
\phantom{}\ce{CH3 - CH = CH - CH2 - C - CH3 -> CH3 - CH = CH - CH2 - C - OH}\phantom{.}
\end{array}\]
Assertion: Aldehydes and ketones, both react with Tollen’s reagent to form silver mirror.
Reason: Both, aldehydes and ketones contain a carbonyl group.
Benzoic acid can be obtained by the oxidation of all of the following EXCEPT ______.
Match List - I with List - II.
| List - I | List - II | ||
| (a) | 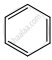 \[\ce{->[CO,HCl][Anhyd. AlCl3/CuCl]}\] \[\ce{->[CO,HCl][Anhyd. AlCl3/CuCl]}\] |
(i) | Hell-Volhard-Zelinsky reaction |
| (b) |
\[\begin{array}{cc}
\ce{O}\phantom{.................}\\ ||\phantom{.................}\\ \ce{R - C - CH3 + NaOX ->} \end{array}\] |
(ii) | Gattermann-Koch reaction |
| (c) | \[\ce{R - CH2 - OH + R'COOH ->[Conc. H2SO4]}\] | (iii) | Haloform reaction |
| (d) | \[\ce{R - CH2COOH ->[(i) X2/Red P][(ii) H2O]}\] | (iv) | Esterification |
Choose the correct answer from the options given below.
Alkaline hydrolysis of C4H8Cl2 gives a compound (A) which on heating with NaOH and I2 produces a yellow precipitate of CHI3. The compound (A) should be ______.
