Advertisements
Advertisements
प्रश्न
Metals are good ______ because they are electron ______.
पर्याय
oxidising agent
reducing agent
acceptors
donors
उत्तर
Metals are good reducing agent because they are electron donors.
संबंधित प्रश्न
An element X belong to 3rd periods and group II of the periodic table state:
whether it is a metal or a non-metal.
iii) Observe the figure and answer the following questions.
a) Identify the block shown by box A and write an electronic configuration of any one element of this block.
b) Identify the block of element denoted by letter B and write its period number.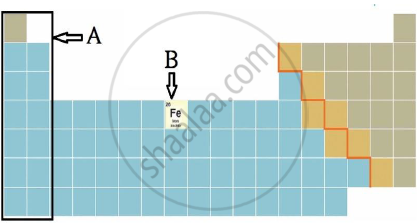
For the main group of the periodic table, the metallic properties of the elements vary approximately with their position as shown in the table.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| H | He | ||||||
| A | B | ||||||
| C | D |
Will the most metallic element be found at A,B,C or D ?
Explain
Larger the atomic size, more metallic is the element.
Supply the missing word from the words given in brackets.
If an element has one electron in its outermost energy level, then it is likely to be _ (metallic, non metallic)
The tendency of an element to form cation is the _______ character of that element.
Write scientific reason.
The metallic character of elements increases while going down the groups.
Write scientific reason.
The non-metallic character increases while going from left to right in a period.
Which of the following sets of atomic numbers belong to that of alkali metals?
Arrange:
Be, Li, C, B, N, O, F (in increasing metallic character).
