Advertisements
Advertisements
प्रश्न
Methyl orange is used as an indicator. It shows colour change in acid, base and neutral substance. Tabulate your result as follows:
| Indicator | Colour change | Inference |
| Methyl Orange | No change | ...................... |
| .................... | Acid | |
| Yellow | ....................... |
उत्तर
| Indicator | Colour change | Inference |
| Methyl Orange | No change | Neutral substance |
| Red | Acid | |
| Yellow | Base |
APPEARS IN
संबंधित प्रश्न
Write the balanced chemical equation for the following reaction.
\[\ce{Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water}\]
Balance the given equation:
H2O2  H2O + O2
H2O + O2
Balance the given equation:
AI(OH)3  AI2O3 +H2O
AI2O3 +H2O
When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are products.
Convey the following information in the form of a balanced chemical equation:
"An aqueous solution of ferrous sulphate reacts with an aqueous solution of sodium hydroxide to form a precipitate of ferrous hydroxide and sodium sulphate remains in solution."
Potassium chlorate (KClO3) on heating forms potassium chloride and oxygen. Write a balanced equation for this reaction and indicate the evolution of gas.
With the help of an appropriate example, justify that some of the chemical reactions are determined by Change in temperature.
Give chemical equation for the reaction involved in the above case.
Give one example of an exothermic reaction.
Which of the following does not involve a chemical reaction?
When the solution of substance X is added to a solution of potassium iodide, then a yellow solid separates out from the solution.
(a) What do you think substance X is likely to be?
(b) Name the substance which the yellow solid consists of.
(c) Which characteristic of chemical reaction is illustrated by this example?
(d) Write a balanced chemical equation for the reaction which takes place. Mention the physical states of all the reactant and products involved in the chemical equation.
A white precipitate will be formed if we add common salt solution to:
(a) Ba(NO3)2 solution
(b) KNO3 solution
(c) AgNO3 solution
(d) Mg(NO3)2 solution
Write word equation for the following skeletal equation:
\[\ce{FeCl2 + Cl2 -> FeCl3}\]
Balance the following equation. Also name the product formed.
`"H"_2 +"CI"_2 → "HCI"`
Write the chemical equation for the following word equation and balance them.
Carbon + Oxygen → Carbon dioxide
Balance the following equation:
Zn + KOH → K2ZnO2 + H2
Balance the following equation:
\[\ce{S + HNO3 -> H2SO4 + NO2 + H2O}\]
Balance the following equation:
P + HNO3 → NO2 + H2O + H3PO4
Balance the following equation:
Zn + HNO3 `→` Zn(NO3)2 + H2O + NO2
Write the balanced chemical equation of the following reaction.
potassium bicarbonate + sulphuric acid → potassium sulphate + carbon dioxide + water
Write the balanced chemical equations of the following reactions.
chlorine + sulphur dioxide + water → sulphuric acid + hydrogen chloride
Write the balanced chemical equation of the following reaction.
copper + nitric acid → copper nitrate + nitric oxide + water
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate mass of MnO2 used.
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate moles of chlorine gas formed.
Considering the relationship in the first pair, complete the second paie :
AgCI : White :: CuI2 : ___________
Write word equation for the following molecular equation:
\[\ce{N2 + 3H2 ⇌[Fe - 450°C][200 atoms] 2NH3 + \triangle}\]
Word equation:
State why [+ Δ] indication is seen after NH3.
State what 200 atmospheres – indicates.
State the function of Fe in the above reaction.
In certain reaction a change of state is observed i.e. solid to liquid, liquid to gas etc. – State the change of state of the products – to give the respective reactant.
NH3 + HCl ⇌ NH4Cl
\[\ce{2KClO3->[MnO2] 2KCl + 3O2[g] - is a balanced equation.}\]
Give a reason why the above equation is balanced.
Balance the following simple equation:
N2 + O2 ⇌ NO
Balance the following simple equation:
Al + N2 → AlN
Balance the following simple equation:
Ca + H2O → Ca(OH)2 + H2
Balance the following simple equation:
Fe + H2O ⇌ Fe3O4 + H2
Write a balanced equation for the following word equation:
Potassium nitrate → Potassium nitrite + Oxygen
Write a balanced equation for the following word equation:
Lead dioxide [lead (IV) oxide] → Lead monoxide + Oxygen
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
Na2SO3 + HCl → 2NaCl + H2O + SO2
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
Fe2O3 + 2H2 → 2Fe + 3H2O
Write an Explanation.
Balanced equations
Balance the given chemical reaction as per the instructions below.
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
Write names of reactants and products of chemical reaction.
Reema took 5ml of Lead Nitrate solution in a beaker and added approximately 4ml of Potassium Iodide solution to it. What would she observe?
Which of the following are exothermic processes?
- Reaction of water with quick lime
- Dilution of an acid
- Evaporation of water
- Sublimation of camphor (crystals)
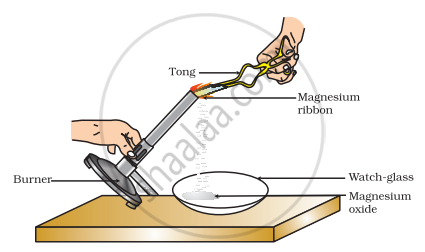
Which of the following is the correct observation of the reaction shown in the above set up?
