Advertisements
Advertisements
प्रश्न
Name the compound formed when ethanol is heated in excess of conc. sulphuric acid at 443 K. Also, write the chemical equation of the reaction stating the role of conc. sulphuric acid in it. What would happen if hydrogen is added to the product of this reaction in the presence of a catalyst such as palladium or nickel?
उत्तर
The compound that is formed when ethanol is heated in excess of conc. sulphuric acid at 443 K is ethene.
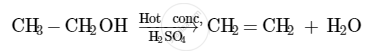
The concentrated sulphuric acid acts as a dehydrating agent in this reaction. It facilitates the removal of a molecule of water from the ethanol molecule.
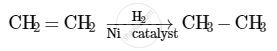
When hydrogen is added to ethene in the presence of a catalyst such as palladium or nickel, saturation of the compound takes place to form ethane.
APPEARS IN
संबंधित प्रश्न
Write the name and structural formula of the compound obtained when ethanol is heated at 443 K with excess of conc. H2SO4. Also write chemical equation for the reaction stating the role of conc. H2SO4 in it.
How would you distinguish experimentally between an alcohol and a carboxylic acid?
What would be observed on adding a 5% alkaline potassium permanganate solution drop by drop to some warm ethanol in a test-tube? Write the name of the compound formed during the chemical reaction. Also write chemical equation of the reaction which takes place.
How is the absolute alcohol obtained?
how can we prepare ethane by reduction of a halogen compound?
Ethane, Ethene, Ethanoic acid, Ethyne, Ethanol
From the compounds given above, name
The compound with -OH as apart of its structure.
Write the uses of ethanol.
How would you bring about the following conversions? Name the process and write the reaction involved.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
