Advertisements
Advertisements
प्रश्न
Name the compound formed when ethanol is heated in excess of conc. sulphuric acid at 443 K. Also, write the chemical equation of the reaction stating the role of conc. sulphuric acid in it. What would happen if hydrogen is added to the product of this reaction in the presence of a catalyst such as palladium or nickel?
उत्तर
The compound that is formed when ethanol is heated in excess of conc. sulphuric acid at 443 K is ethene.
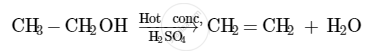
The concentrated sulphuric acid acts as a dehydrating agent in this reaction. It facilitates the removal of a molecule of water from the ethanol molecule.
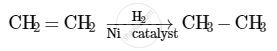
When hydrogen is added to ethene in the presence of a catalyst such as palladium or nickel, saturation of the compound takes place to form ethane.
APPEARS IN
संबंधित प्रश्न
Give the structural formula of ethanol.
Write a balanced chemical equations for Heating of Ethanol at 170°C in the presence of conc. Sulphuric acid
The catalyst used in the contact process is
(A) Copper
(B) Iron
(C) Vanadium pentoxide
(D) Manganese dioxide
State the method of preparation of ethanol by hydrolysis of ethyl bromide.
What would you see, when ethyne is bubbled through a solution of bromine in carbon tetrachloride?
[Ethane, Ethene, Ethanoic acid, Ethyne, Ethanol]
From the above, name the homologue of the homologous series with the general formula CnH2n.
State how the following conversion can be carried out:
Ethene to ethyl alcohol
______ react with dil. HNO3 to evolve hydrogen gas.
Name the compound formed when ethanol is heated at 443 K in the presence of conc. \[\ce{H2SO4}\] and draw its electron dot structure.
State the role of conc. \[\ce{H2SO4}\] in the reaction.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
