Advertisements
Advertisements
प्रश्न
Observe the figure and write the answers to the following questions.

- Write the name of the reaction shown in the following figure.
- Write the above chemical reaction in the form of a balanced equation.
- Write the name of the product produced in the above reaction, write a use.
- Write the name of the catalyst used in the above reaction.
उत्तर
- The reaction shown in the figure is esterification.
- Balanced equation of the reaction between ethanol and ethanoic acid is as follows:
\[\ce{\underset{\text{Ethanol}}{C2H5OH} + \underset{\text{Ethanoic acid}}{CH3COOH} ->[Conc.][H2SO4] \underset{\text{Ethyl ethanoate}}{CH3COOC2H5} + \underset{\text{Water}}{H2O}}\] - The product formed is ethyl ethanoate.
Use: It is used for making fragrances and flavouring agents. - The catalyst used in the reaction is concentrated sulphuric acid.
APPEARS IN
संबंधित प्रश्न
Consider the following comments about saponification reactions:
I. Heat is evolved in these reactions.
II. For quick precipitation of soap, sodium chloride is added to the reaction mixtures.
III. Saponification reactions are a special kind of neutralisation reactions.
IV. Soaps are basic salts of long-chain fatty acids.
The correct comments are
(a) I, II and III
(b) II, III and IV
(c) I, II and IV
(d) Only I and IV
A gas is liberated immediately with a brisk effervescence, when you add acetic acid to sodium hydrogen carbonate powder in a test tube. Name the gas and describe the test that confirms the identity of the gas.
Give the common names and IUPAC names of the following compounds of CH3COOH.
Fill in the following blank with suitable word:
The next higher homologue of ethanol is ...............
Complete the following equation:
`CH_3 COOH + CH_2 H_5 OH` 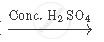
Fill in the following blank with a suitable word:
The organic acid present in vinegar is ______.
What type to compound is CH3COOH?
An organic compound X of molecular formula C2H4O2 gives brisk effervescence with sodium hydrogen carbonate. Give the name and formula of X.
When ethanoic acid reacts with sodium hydrogen carbonate, then a salt X is formed and a gas Y is evolved. Name the salt X and gas. Y Describe an activity with the help of a labelled diagram of the apparatus used to prove that the evolved gas is the one which you have named. Also write the chemical equation of the reaction involved.
Give any two uses of ethanoic acid.
A neutral organic compound is warmed with some ethanoic acid and a little of conc. H2SO4. Vapours having sweet smell (fruity smell) are evolved. What type of functional group is present in this organic compound?
Acetic acid is a typical acid. Write one equation in case of its reactions with a metal?
Acetic acid is a typical acid. Write one equation in case of its reactions with a bicarbonate.
What do you observe when acetic acid is added to ethyl alcohol in the presence of sulphuric acid?
Write a balanced chemical equation for the following:
A mixture of sodalime and sodium acetate is heated.
State the observation
When the gaseous product obtained by dehydration of ethyl alcohol is passed through bromine water.
A compound X when treated with an organic acid Y (having vinegar-like smell) in the presence of the acid Z, forms a compound P which has a fruity smell.
- Identify X, Y, and Z
- Write the structural formula of X and Y.
- What type of compound P is?
- Name the above reaction.
- If compound X and Y both have 2 carbon atoms. Write the reaction.
Two statements are given - one labelled Assertion (A) and the other labelled Reason (R).
Assertion (A): Esterification is a process in which a sweet-smelling substance is produced.
Reason (R): When esters react with sodium hydroxide an alcohol and sodium salt of carboxylic acid are obtained.
Why is glacial acetic acid called so?
A spatula full of sodium carbonate is taken in a test tube and 2 mL of dilute ethanoic acid is added to it.
Write a chemical equation for the reaction.
