Advertisements
Advertisements
प्रश्न
Predict the co-ordination No. of cs+ ion if `r_(Cs)^+` = 1.69Å and `r_(Cl)^-` = 1.81Å.
उत्तर
The radius ratio (r+/r–) defines the coordination number of the cation
`r_(Cs^+) = 1.69 Å, r_(Cl^-) = 1.81 Å`
`∴ r_(Cs^+)/r_(Cl^-) = 1.69/1.89 = 0.9337`
Since, radius ratio is greater than 0.732, the coordination number of cation (Cs+) is 8.
APPEARS IN
संबंधित प्रश्न
Classify the following ligands into monodentate and polydentate —
- Ammonia
- Carbon monoxide
- Ethylene diamine
- Ethylene diamine tetra acetate ion
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
cis-[CrCl2(en)2]Cl
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
[Mn(H2O)6]SO4
Write structures of compounds A, B and C in of the following reactions
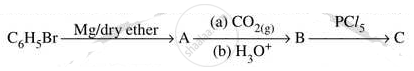
Write structures of compounds A, B and C in of the following reactions
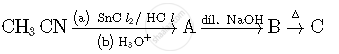
Following compounds are given to you :
2-Bromopentane, 2-Bromo-2-methylbutane, 1-Bromopentane
1) Write the compound which is most reactive towards SN2 reaction.
2) Write the compound which is optically active.
3) Write the compound which is most reactive towards β-elimination reaction.
Write IUPAC name of the following Complex [Cr(NH3)3Cl3]
Write the structures of compounds A, B and C in the following reactions
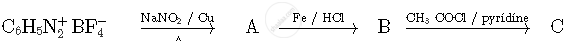
A group of atoms can function as a ligand only when:
Which of the following is non-ionizable?
The correct \[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2Cl2]}\] is ______.
Which of the following complexes are homoleptic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Co(NH3)4 Cl2]^{+}}\]
(iii) \[\ce{[Ni(CN)4]^{2-}}\]
(iv) \[\ce{[Ni(NH3)4Cl2]}\]
Match the complex ions given in Column I with the colours given in Column II and assign the correct code:
| Column I (Complex ion) | Column II (Colour) |
| A. \[\ce{[Co(NH3)6]^{3+}}\] | 1. Violet |
| B. \[\ce{[Ti(H2O)6]^{3+}}\] | 2. Green |
| C. \[\ce{[Ni(H2O)6]^{2+}}\] | 3. Pale blue |
| D. \[\ce{(Ni(H2O)4 (en)]^{2+} (aq)}\] | 4. Yellowish orange |
| 5. Blue |
What is the relationship between observed colour of the complex and the wavelength of light absorbed by the complex?
In which of the following compounds, the oxidation number of iodine is fractional?
Which one of the following ligands forms a chelate?
Ethylene diamine tetraacetate (EDTA) ion is ______.
Why chelate complexes are more stable than complexes with unidentate ligands?
Given below are two statements.
- Statement I: In CuSO4 · 5H2O, Cu-O bonds are present.
- Statement II: In CuSO4 · 5H2O, ligands coordinating with Cu(II) ion are O- and S based ligands.
In light of the above statement, choose the correct answer from the options given below.
The correct order of the spin-only magnetic moment of metal ions in the following low spin complexes,
\[\ce{[V(CN)6]^{4-}, [Fe(CN)6]^{4-}, [Ru(NH3)6]^{3+} and [Cr(NH3)6]^2}\] is:
What is a chelate complex? Give one example.
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
What is meant by the chelate effect? Give an example.
Explain the following, giving two examples:
Ligand
Explain the following, giving two examples:
Coordination polyhedron
Give two examples of unidentate ligand.
What is meant by didentate ligand?
