Advertisements
Advertisements
प्रश्न
Predict the possible product of the following reaction.
nitration of toluene
उत्तर
The Methyl group is the ortho and para directing group. So, nitration of toluene gives p-nitrotoluene and o-nitrotoluene.
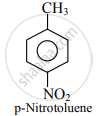
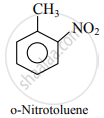
APPEARS IN
संबंधित प्रश्न
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify A, B, C in the following reaction sequence:
\[\ce{CH3 - CH = CH2 ->[Br2/CCl4][room temperature] A ->[Zn] B ->[dil. alkaline][KMnO4] C}\]
Identify giving reason whether the following compound is aromatic or not.

Identify the compound (A) in the following reaction
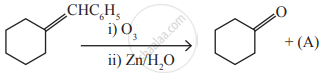
Consider the nitration of benzene using mixed con H2SO4 and HNO3 if a large quantity of KHSO4 is added to the mixture, the rate of nitration will be ______.
An alkane is obtained by decarboxylation of sodium propionate. Same alkane can be prepared by ______.
Identify the compound A, B, C and D in the following series of reactions.

Describe the mechanism of Nitration of benzene.
Write the chemical equations for combustion of propane.
Suggest the route for the preparation of the following from benzene.
m - dinitro benzene
What happens when Isobutylene is treated with acidified potassium permanganate?
Write the structure of the following alkanes.
2, 3 – Dimethyl – 6 – (2 – methyl propyl) decane

Identify A and B.
When acetylene is passed through red hot iron tube at 873 K, ____________ is/are obtained as product/s.
Identify the hydrocarbon compound from following containing carbon atoms in the range of C6 to C8?
−Cl group is ____________.
Which of the following is INCORRECT for aromatic hydrocarbons?
The alkane formed on heating sodium butanoate with sodalime is ______.
Which of the following molecules has shortest C - C bond length?
Arenes on treatments with chlorine in the presence of ferric chloride as a catalyst undergo what type of reaction?
Identify the X and Y in the following reactions.
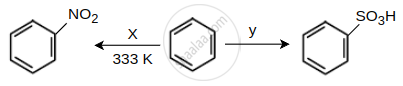
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{.............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{...............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
