Advertisements
Advertisements
प्रश्न
Prove the Mayer's relation `C_p - C _v = R/J`
उत्तर
Consider one mole of an ideal gas that is enclosed in a cylinder by a light, frictionless, airtight piston.
Let P, V, and T be the pressure volume and temperature of the gas, respectively. If the gas is heated so that its temperature rises by dT, but volume remains constant.
Let dQ1 → be the amount of heat supplied to the gas,
dE → be the increase in internal energy. Since, volume of gas is constant, no work is done in moving the piston.
dQ1 = dE + dW
∴ dQ1 = dE = CVdT ...(i)
(As dW = 0)
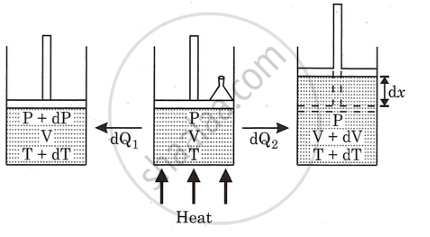
If the gas is heated to the same temperature at constant pressure.
Let dV → be the increase in volume.
Therefore work is done and it is given by
dW = PdV
Let dQ2 → be the heat supplied to the gas
∴ dQ2 = dE + dW
∴ CpdT = dE + dW ...(ii)
Putting dE = CvdT and dW = PdV
∴ Equation (ii) becomes
CPdT = CvdT + PdV
(Cp − Cv)dT = PdV ...(iii)
For one mole of gas
PV = RT
∴ pdV = RdT
∴ (Cp − Cv)dT = RdT
Cp − Cv = R ...(iv)
This is known as Mayer's relation.
Generally, heat supplied is measured in calories, and work done is measured in joules. Therefore, equation (iv) is modified as follows:.
`C_p - C _v = R/J`
APPEARS IN
संबंधित प्रश्न
Calculate the mass of ice required to lower the temperature of 300 g of water 40°C to water at 0°C.
(Specific latent heat of ice = 336 J/g, the Specific heat capacity of water = 4.2J/g°C)
Water in lakes and ponds do not freeze at once in cold countries. Give a reason is support of your answer.
Heat supplied to a solid change it into liquid. What is this change in the phase called?
Differentiate between heat capacity and specific heat capacity.
Name a liquid which has the highest specific heat capacity.
Write the approximate value of specific heat capacity of water in S.I. unit.
What do you mean by the following statement?
The heat capacity of a body is 50 JK-1?
Two metallic blocks P and Q of masses in ratio 2: 1 are given the same amount of heat. If their temperature rise by the same amount, compare their specific heat capacities.
Give one example where high specific heat capacity of water is used as cooling purposes?
Describe a method to determine the specific heat capacity of a solid, like a piece of copper ?
It is generally cold after a hail-storm then during and before the hail storm. Give reason.
Name two green house gases ?
What impact will global warming have on the health of the affected population?
The specific heat capacity of a body depends on _____________ .
650 J of heat is required to raise the temp. of 0.25 kg of lead from 15°C to 35°C. Calculate the Sp. heat capacity of lead.
Fill in the following blank using suitable word:
SI unit of heat is .........
Explain, why is water sprayed on roads in evening in hot summer?
Explain, why does a wise farmer water his fields, if forecast is forst?
An equal quantity of heat is supplied to two substances A and B. The substance A shows a greater rise in temperature. What can you say about the heat capacity of A as compared to that of B?
1 kg of water freezes to form ice at 0°C. What amount of heat is withdrawn?
63.2 g of copper at 50°C can just melt 3.8g of ice. If the specific latent heat of ice is 336 J/g, find the specific heat capacity of copper.
State factors on which the amount of heat radiated by a body depends.
For a gas, `"R"/"C"_"v"=0.4`, where R Is universal gas constant and Cv is the molar specific heat at constant volume. The gas is made up of molecules, which are ______
Two uniform brass rods A and B of length land 2l and radii 2r and r respectively are heated to the same temperature. The ratio of the increase in the volume ofB to that of A is ____________.
For a gas `"R"/"C"_"v" = 0.4,` where 'R' is the universal gas constant and 'Cv' is molar specific heat at constant volume. The gas is made up of molecules which are ______.
Two metals A and B have specific heat capacities in the ratio 2:3. If they are supplied same amount of heat then
Which metal piece will have greater mass if the rise in temperature is the same for both metals?
The specific heat capacity of ______ is maximum.
Specific heat capacity of a substance X is 1900 Jkg-1°C-1 means ______.
