Advertisements
Advertisements
Question
Prove the Mayer's relation `C_p - C _v = R/J`
Solution
Consider one mole of an ideal gas that is enclosed in a cylinder by a light, frictionless, airtight piston.
Let P, V, and T be the pressure volume and temperature of the gas, respectively. If the gas is heated so that its temperature rises by dT, but volume remains constant.
Let dQ1 → be the amount of heat supplied to the gas,
dE → be the increase in internal energy. Since, volume of gas is constant, no work is done in moving the piston.
dQ1 = dE + dW
∴ dQ1 = dE = CVdT ...(i)
(As dW = 0)
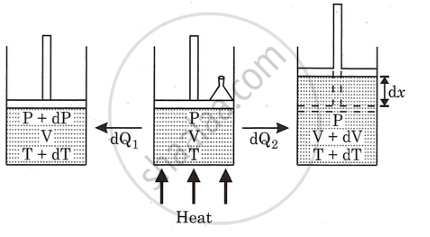
If the gas is heated to the same temperature at constant pressure.
Let dV → be the increase in volume.
Therefore work is done and it is given by
dW = PdV
Let dQ2 → be the heat supplied to the gas
∴ dQ2 = dE + dW
∴ CpdT = dE + dW ...(ii)
Putting dE = CvdT and dW = PdV
∴ Equation (ii) becomes
CPdT = CvdT + PdV
(Cp − Cv)dT = PdV ...(iii)
For one mole of gas
PV = RT
∴ pdV = RdT
∴ (Cp − Cv)dT = RdT
Cp − Cv = R ...(iv)
This is known as Mayer's relation.
Generally, heat supplied is measured in calories, and work done is measured in joules. Therefore, equation (iv) is modified as follows:.
`C_p - C _v = R/J`
APPEARS IN
RELATED QUESTIONS
A geyser heats water flowing at the rate of 3.0 litres per minute from 27 °C to 77 °C. If the geyser operates on a gas burner, what is the rate of consumption of the fuel if its heat of combustion is 4.0 × 104 J/g?
A copper vessel of mass 100 g contains 150 g of water at 50°C. How much ice is needed to cool it to 5°C?
Given: Specific heat capacity of copper = 0.4 Jg-1 °C-1
The Specific heat capacity of water = 4.2 Jg-1 °C-1
The Specific latent heat of fusion ice = 336 Jg-1
Specific heat capacity of substance A is 3.8 J g-1K-1 whereas the specific heat capacity of substance B is 0.4 J g-1 K-1
(i) Which of the two is a good conductor of heat?
(ii) How is one led to the above conclusion?
(iii) If substances A and B are liquids then which one would be more useful in car radiators?
Write the expression for the heat energy Q received by the substance when m kg of substance of specific heat capacity c Jkg-1 k-1 is heated through Δt° C.
Discuss the role of high specific heat capacity of water with reference to climate in coastal areas.
Water is used in hot water bottles for fomentation. Give a reason.
Explain the term boiling ?
Name three fossil fuels that emit carbon dioxide into the atmosphere ?
How will rise in sea level affect population in coastal countries?
A substance is heated at a constant rate from a low temperature to a high temperature. A graph of temperature against time is shown in the figure. Which part or parts of the graph correspond(s) to the substance existing in two states?

(b) 2000 J of heat energy is required to raise the temperature of 4 kg of a
metal by 3°c. Which expression gives the specific heat capacity of the metal?
Define heat capacity and state its units.
A burner raises the temperature of 360 g of water from 40°C to 100°C in 5 minutes. Calculate the rate of heat supplied by the burner.
State, with reason, which of the two, boiling water or steam both at 100°C will produce more severe burns.
What is heat? What is the S. I. unit of heat?
What are the factors on which the quantity of heat given to a body depends?
1 kg of water freezes to form ice at 0°C. What amount of heat is withdrawn?
A substance is in the form of a solid at 0°C. The amount of heat added to this substance and the temperature of the substance are plotted on the following graph:

If the specific heat capacity of the solid substance is 500 J/kg °G, find from the graph, the mass of the substance.
A. hot solid of mass 60 g at 100°C is placed in 150 g of water at 20° C. The final steady temperature recorded is 25°C. Calculate the specific heat capacity of the solid. [Specific heat capacity of water = 4200 J kg-1 °C-1]
Calculate the amount of heat released when 5.0 g of water at 20°C is changed into ice at 0°C.
(Specific heat capacity of water = 4.2 J/g°C
Specific latent heat of fusion of ice = 336 J/g)
Numerical Problem.
What could be the final temperature of a mixture of 100 g of water at 90 °C and 600g of water at 20°C.
Derive Meyer’s relation for an ideal gas.
The molar specific heats of an ideal gas at constant pressure and constant volume are denoted by Cp and Cv respectively. If `gamma = "C"_"p"/"C"_"v"` and R is the universal gas constant, then Cp is equal to ______.
A monoatomic gas of pressure 'P' having volume 'V' expands isothermally to a volume '2V' and then adiabatically to a volume '16V'. The final pressure of the gas is ______.
(ratio of specific heats = `5/3`)
An office room contains about 4000 moles of air. The change in the internal energy of this much air when it is cooled from 34° C to 19° C at a constant pressure of 1.0 atm is (Use `gamma_"air"` = 1.4 and Universal gas constant = 8.314 J / mol K) ____________.
Two metals A and B have specific heat capacities in the ratio 2:3. If they are supplied same amount of heat then
Which metal piece will have greater mass if the rise in temperature is the same for both metals?
Two metals A and B have specific heat capacities in the ratio 2:3. If they are supplied same amount of heat then
If specific heat capacity of metal A is 0.26 Jg-1 0C-1 then calculate the specific heat capacity of metal B.
Water has the lowest specific heat capacity.
