Advertisements
Advertisements
प्रश्न
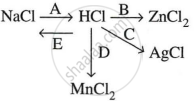
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
उत्तर
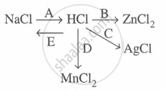
A: \[\ce{NaCl + H2SO4 ->[< 200°C] NaHSO4 + HCl ^}\]
B: \[\ce{Zn + 2HCl -> ZnCl2 + H2}\]
C: \[\ce{AgNO3 + HCl -> AgCl + HNO3}\]
D: \[\ce{MnO2 + 4HCl ->[\Delta] MnCl2 + 2H2O + Cl2 ^}\]
E: \[\ce{HCl + NaOH -> NaCl + H2O}\]
APPEARS IN
संबंधित प्रश्न
Hydrochloric acid contains (i) hydrogen (ii) chlorine. Prove it. Write equations for the reactions.
Name a black metallic oxide which reacts with hydrochloric acid to give a coloured solution.
Name the chemical in which gold can be dissolved.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{Pb(NO3)2 + HCl ->}\]
Complete the following reaction and balance them.
Zn+ 2HCl ⟶
Calcium oxide and phosphorous pentoxide are very good drying agents but they are not used to dry HCI gas. Give reasons for each.
Give a balanced equation when dilute hydrochloric acid is added to : Lead (II) sulphide
Complete and balance the following reaction, state whether dilute or conc. acid is used.
NH4OH + HCl ⟶
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
