Advertisements
Advertisements
प्रश्न
The work function of a metal is 2.31 eV. Photoelectric emission occurs when the light of frequency 6.4 × 1014 Hz is incident on the metal surface. Calculate
- the energy of the incident radiation,
- the maximum kinetic energy of the emitted electron and
- the stopping potential of the surface.
उत्तर
(i) Frequency of incident radiation = ν = 6.4 × 1014 Hz
Energy of incident radiation = E = hν = 6.6 × 10−34 × 6.4 × 1014 = 42.24 × 10−20 J
(ii) KEmax = hν − Φ0
∴ KEmax = 42.24 × 10−20 − 2.31 × 1.6 × 10−19 = 5.28 × 10−20 J
(iii) If stopping potential = VS, then
eVS = KEmax
∴ VS = `"KE"_"max"/"e"`
= `(5.28 xx 10^-20)/(1.6 xx 10^-19)`
= 3.3 × 10−1
= 0.33 V
APPEARS IN
संबंधित प्रश्न
Find the maximum frequency of X-rays produced by 30 kV electrons.
How will the thermionic current vary if the filament current is increased?
An isolated metal sphere is heated to a high temperature. Will it become positively charged due to thermionic emission?
Why does thermionic emission not take place in non-conductors?
The work function of aluminum is 4⋅2 eV. If two photons each of energy 2⋅5 eV are incident on its surface, will the emission of electrons take place? Justify your answer.
The wavelength λe of an electron and λp of a photon of same energy E are related by
Why do metals have a large number of free electrons?
Photoelectric emission is observed from a metallic surface for frequencies ν1 and ν2 of the incident light (ν1 > ν2). If the maximum value of kinetic energy of the photoelectrons emitted in the two cases are in the ration 1 : n then the threshold frequency of the metallic surface is ______.
Consider Figure for photoemission.
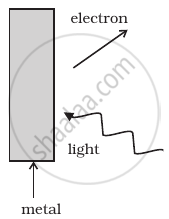
How would you reconcile with momentum conservation? Note light (photons) have momentum in a different direction than the emitted electrons.
Name the factors on which photoelectric emission from a surface depends.
