Advertisements
Advertisements
प्रश्न
What happens when a solution of an acid is mixed with a solution of a base in a test tube?
- The temperature of the solution increases
- The temperature of the solution decreases
- The temperature of the solution remains the same
- Salt formation takes place
पर्याय
(i) only
(i) and (iii)
(ii) and (iii)
(i) and (iv)
उत्तर
(i) and (iv)
Explanation -
Salt formation takes place in the neutralisation reaction. It is always exothermic and temperature increases.
APPEARS IN
संबंधित प्रश्न
Why should curd and sour substances not be kept in brass and copper vessels?
Name one natural source of the following acids:
Lactic acid
Write the chemical formula of soda ash?
What is the chemical name of bleaching powder?
With the help of suitable examples explain the term neutralization and the formation of –
- acidic,
- basic and
- neutral salts
Which of the following salts does not contain any water of crystallisation?
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
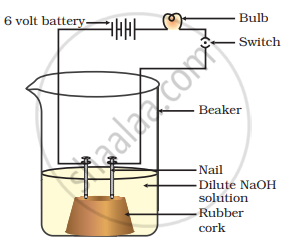
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
Vinegar is ______ in taste.
Take a clean test tube with a holder and pour some dilute hydrochloric acid. Add a few pieces of magnesium ribbon pieces slowly. What do you observe? Now show a burning match stick near the mouth of the test tube. Do you hear any sound? The gas burns with a pop sound. From this, it is observed that hydrogen gas has been formed due to the reaction between acid and metal.
Two salts 'X' and 'Y' are dissolved in water separately. When phenolphthalein is added to these two solutions, the solution 'X' turns pink and the solution 'Y' does not show any change in colour, therefore 'X' and 'Y' are:
