Advertisements
Advertisements
प्रश्न
What happens when methanal undergoes cannizzaro reaction?
उत्तर १
When methanal (formaldehyde, HCHO) undergoes the Cannizzaro reaction, it results in the formation of formic acid HCOOH and methanol CH3OH.
The Cannizzaro reaction is a base-catalyzed disproportionation reaction that occurs with non-alpha-hydrogenated aldehydes (like methanal). In this reaction, one molecule of formaldehyde gets reduced to methanol CH3OH, while the other gets oxidized to formic acid HCOOH.
The reaction can be represented as:
\[\ce{2HCHO ->[NaOH] HCOOH + CH3OH}\]
उत्तर २
Methanal will undergo Cannizzaro reaction as it possesses α-hydrogen to form methanol.
\[\begin{array}{cc}
\phantom{.}\ce{H}\phantom{..............................}\ce{H}\phantom{.......}\ce{H}\phantom{...........}\phantom{..}\\
\backslash\phantom{..............................}|\phantom{.........}\backslash\phantom{..........}\\
\ce{2C = O + conc.KOH -> H - C - OH + C - O- K+}\\
/\phantom{..............................}|\phantom{.........}/\phantom{..........}\\
\ce{\underset{Methanal}{H}}\phantom{.........................}\ce{\underset{Methanol}{H}}\phantom{.}\ce{\underset{methanoate}{\underset{Potassium}{H}}}\phantom{............}\\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Write a note on the self oxidation-reduction reaction of an aldehyde with a suitable example.
An organic compound with the molecular formula C9H10O forms 2, 4-DNP derivative, reduces Tollens’ reagent and undergoes Cannizzaro reaction. On vigorous oxidation, it gives 1, 2-benzenedicarboxylic acid. Identify the compound.
How will you convert acetone to acetone cyanohydrin?
Write the product formed when p-nitro chlorobenzene is heated with aqueous NaOH at 443K followed by acidification?
complete the following reaction:
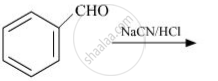
Complete the following reaction:

The products obtained in the Cannizzaro reaction are
The key step in cannizzaro reaction in the inter molecular shift qf
In the Cannizzaro reaction given below:
\[\ce{2Ph-CHO ->[OH^-] Ph-CH2OH + PhC\overset{-}{O}_2}\]
the slowest step is:
Convert the following:
Benzene to m-nitrobenzaldehyde
