Advertisements
Advertisements
प्रश्न
What is meant by a reversible and irreversible processes?
उत्तर
Reversible processes: A thermodynamic process can be considered reversible only if it possible to retrace the path in the opposite direction in such a way that the system and surroundings pass through the same states as in the initial, direct process.
Irreversible processes: All natural processes are irreversible. Irreversible processes cannot be plotted in a PV diagram, because these processes cannot have unique values of pressure, the temperature at every stage of the process.
APPEARS IN
संबंधित प्रश्न
Explain why The climate of a harbour town is more temperate than that of a town in a desert at the same latitude.
3 mole of a gas at temperature 400 K expands isothermally from an initial volume of 4 litres to a final volume of 8 litres. Find the work done by the gas. (R = 8.31 J mol-1 K-1)
Explain graphically (i) positive work with varying pressure, (ii) negative work with varying pressure, and (iii) positive work at constant pressure.
Write a note on free expansion.
When a cycle tyre suddenly bursts, the air inside the tyre expands. This process is ____________.
Give the equation of state for an adiabatic process.
Consider the following cyclic process consist of isotherm, isochoric and isobar which is given in the figure.
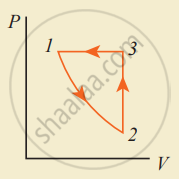
Draw the same cyclic process qualitatively in the V-T diagram where T is taken along the x-direction and V is taken along the y-direction. Analyze the nature of heat exchange in each process.
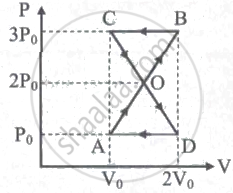
A thermodynamic system undergoes cyclic process ABCDA as shown in the figure. The work done by the system is ______
The work done on the system in changing the state of a gas adiabatically from equilibrium state A to equilibrium state B is 22.4 J. If the gas is taken from state A to B through another process in which the net heat absorbed by the system is 15.5 cal, then the net work done by the system in the latter case is ______.
( l cal = 4.2 J)
Give any two types of a thermodynamic process.
