Advertisements
Advertisements
प्रश्न
What is the action of following on phenol at low temperature?
- dil. HNO3
- conc. H2SO4
- Br2/CS2
उत्तर
- Dilute HNO3: Phenol reacts with dilute nitric acid at low temperature to give a mixture of ortho- and para-nitrophenol.
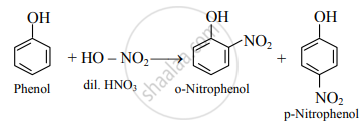
- Conc. H2SO4: At room temperature (298 K), phenol reacts with concentrated sulphuric acid to form o-phenolsulphonic acid.
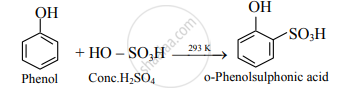
- Br2 in CS2: When a reaction is carried out in a solvent of lower polarity like CS2 a mixture of ortho- and para-bromophenol is formed.

संबंधित प्रश्न
Answer in brief.
Explain why p-nitrophenol is a stronger acid than phenol.
An ether (A), C5H12O, when heated with excess of hot HI produce two alkyl halides which on hydrolysis form compound (B) and (C), oxidation of (B) gave and acid (D), whereas oxidation of (C) gave a ketone (E). Deduce the structural formula of (A), (B), (C), (D), and (E).
When vapours of tert.butyl alcohol are passed over hot copper, it gives _____________
Reaction between hot conc. HI and anisole gives ______________
Write the reaction to get aspirin from salicylic acid.
What is the action of following reagents on pent-3-enal?
- H2/Ni
- LiAlH4/H3O+
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
Write chemical equation of acetyl chloride with phenol
How will you bring about the following conversions?
2-methyl propan-2-ol to 2-methylpropene
Phenol reacts with concentrated nitric acid in the presence of cone. H2SO4 to form ____________.
\[\ce{C3H8O ->[KMnO4][(Oxidation)] C3H6O2}\]
The compound C3H8O is a/an ____________.
In the Lucas test for alcohols, the appearance of turbidity is due to the formation of ____________.
Which of the following compounds does not react with bromine in alkaline medium?
Phenol is ____________.
Cumene is used in the commercial method for the manufacture of ____________.
Carbolic acid is oxidised by acidified sodium dichromate to give ______.
____________ will NOT undergo acetylation.
Phenoxide ion is more stable than phenol due to the ____________.
The most resistant alcohol towards oxidation reaction is:
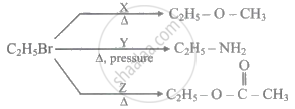
Identify reagents X, Y and Z.
Which of the following reagents is used to convert phenol to benzene?
Identify the product B in following conversion.
\[\ce{Chlorobenzene + H2O ->[Cu, 673 K][Pressure] A ->[conc. H2SO4][373 K] B}\]
Identify 'Z' in the following series of reaction:
\[\ce{Butan - 2 - ol ->[PCl3] X ->[alco. KOH] Y ->[i) H2SO4][ii) H-OH/heat] Z}\]
Which of the following on oxidation yields ethyl methyl ketone?
Which among the following phenolic compound is most acidic in nature?
Identify the compound amongst the following of which 0.1 M aqueous solution has highest boiling point.
The acid, which contains both -OH and -COOR groups is ______.
Which among the following is not the method of preparation of phenol?
The chemical test that distinguish between benzoic acid and phenol is ______.
Write the chemical reaction when hot copper is treated with Vapours of 2° (secondary) alcohol.
