Advertisements
Advertisements
प्रश्न
Which of the following is not a straight chain hydrocarbon?
पर्याय
\[\begin{array}{cc}
\ce{H3C - CH2 - CH2 - CH2 - CH2}\\
\phantom{.........................}|\\
\phantom{............................}\ce{CH3}
\end{array}\]\[\ce{H3C - CH2 - CH3 - CH2 - CH3}\]
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{H2C - H2C - H2C - CH2}\\
\phantom{...................}|\\
\phantom{.....................}\ce{CH3}
\end{array}\]\[\begin{array}{cc}
\ce{CH3}\phantom{.............................}\\
\backslash\phantom{.........................}\\
\ce{CH - CH2 - CH2 - CH3}\\
/\phantom{.........................}\\
\ce{H3C}\phantom{.............................}\\
\end{array}\]
उत्तर
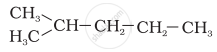
Explanation -
In the hydrocarbon (d), branching takes place at carbon-2.
APPEARS IN
संबंधित प्रश्न
Write answer as directed.
Give any four functional groups containing oxygen as the heteroatom in it. Write name and structural formula of one example each.
Define Hydrocarbons.
Find the odd one out and give its explanation.
Cyclohexane is a branched chain type of hydrocarbon.
In LPG, butane is a flammable component.
Explain the concept of heteroatoms with the help of examples.
Distinguish between:
Saturated hydrocarbons - Unsaturated hydrocarbons
Consider the following organic compounds:
| (i) \[\begin{array}{cc} \ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\ |\phantom{....}|\phantom{....}|\\ \ce{H - C - C - C = O}\\ |\phantom{....}|\phantom{.....}\\ \ce{H}\phantom{....}\ce{H}\phantom{.....} \end{array}\] |
(ii) \[\begin{array}{cc} \phantom{....}\ce{H}\phantom{...}\ce{H}\phantom{....}\\ \phantom{....}|\phantom{....}|\phantom{....}\\ \ce{H - C - C = O}\\|\phantom{.....}\\ \ce{H}\phantom{.....}\end{array}\] |
- Name the functional group present in these compounds.
- Write the general formula for the compounds of this functional group.
- State the relationship between these compounds and draw the structure of any other compound having a similar functional group.
Identify heteroatom (s) in the following compound.
\[\begin{array}{cc}
\ce{CH3CH2 - C - CH3}\\\
\phantom{....}||\
\ce\\\phantom{....}{O}\end{array}\]
One variety of household fuel is a mixture of propane (60%) and butane (40%). If 20 litres of this mixture is burnt, find the total volume of carbon dioxide added to the atmosphere. The combination reactions can be represented as:
\[\ce{C3H8 + 5O2 -> 3CO2 + 4H2O}\]
\[\ce{2C4H10 +13O2 -> 8CO2 + 10H2O}\]
