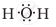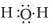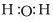Advertisements
Advertisements
प्रश्न
The correct electron dot structure of a water molecule is
पर्याय
उत्तर

Explanation -
The central atom in water molecule is oxygen. It has 6 electrons in its valence shell and shares one electron each from the two hydrogen atoms.
APPEARS IN
संबंधित प्रश्न
How many structural isomers can you draw for pentane?
Fill in the following blank with suitable word:
Ethyne has ......... carbon-hydrogen single bonds.
Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds:
ethene
How many isomers of the following hydrocharbons are possible?
C3H8
What happens when vegetable oils are hydrogenated? Name the catalyst used.
What is added to groundnut oil when it is to be converted to vanaspati ghee?
Which of the following hydrocarbons can decolourise bromine water and which cannot? Why?
C6H12, C6H14, C6H10
Structural formula of ethyne is ______.
Identify the unsaturated compounds from the following:
- Propane
- Propene
- Propyne
- Chloropropane
The formulae of four organic compounds are shown below. Choose the correct option
| A | B | C | D |
| \[\begin{array}{cc} \phantom{.}\ce{H}\phantom{......}\ce{H}\\ \phantom{.}\backslash\phantom{.....}/\\ \ce{C = C}\\ /\phantom{.....}\backslash\\ \ce{H}\phantom{......}\ce{H} \end{array}\] |
\[\begin{array}{cc} \phantom{........}\ce{H}\phantom{.....}\ce{O}\\ \phantom{.......}|\phantom{....}//\\ \ce{H - C - C}\\ \phantom{......}|\phantom{.....}\backslash\\ \phantom{...........}\ce{H}\phantom{.....}\ce{O - H} \end{array}\] |
\[\begin{array}{cc}
|
\[\begin{array}{cc} \ce{H}\phantom{...}\ce{H}\phantom{....}\\ |\phantom{....}|\phantom{....}\\ \ce{H - C - C - O - H}\\ |\phantom{....}|\phantom{.....}\\ \ce{H}\phantom{....}\ce{H}\phantom{.....} \end{array}\] |