Advertisements
Advertisements
प्रश्न
Write down functional isomers of a carbonyl compound with molecular formula \[\ce{C3H6O}\]. Which isomer will react faster with \[\ce{HCN}\] and why? Explain the mechanism of the reaction also. Will the reaction lead to the completion with the conversion of whole reactant into product at reaction conditions? If a strong acid is added to the reaction mixture what will be the effect on concentration of the product and why?
उत्तर
\[\ce{C3H6O}\] will have following isomer.
\[\ce{\underset{Proponal (I)}{CH3CH2CHO}}\]
\[\begin{array}{cc}
\phantom{}\ce{O}\\
\phantom{}||\\
\ce{\underset{Propan-2-one (II)}{CH3 - C - CH3}}
\end{array}\]
Compound I will react faster with HCN due to less steric hindrance and greater positive charge on carbon atom of carbonyl group. Two methyl groups increase electron density on carbonyl carbon in compounds II hence the rate of nucleophilic attack is less.
Mechanism of the reaction:
\[\ce{HCN + OH- -> CN- + H2O}\]
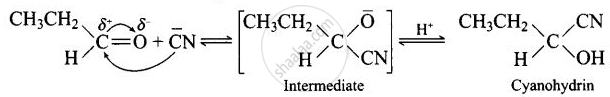
The reaction will not lead to completion since it is a reversible reaction, hence equilibrium is established.
If a strong acid is added to reaction mixture, the addition is inhibited because the formation of \[\ce{CN}\]” ions form \[\ce{HCN}\] is prevented.
APPEARS IN
संबंधित प्रश्न
Arrange the following in the increasing order of their boiling points : C2H5OH, CH3 − CHO, CH3 − COOH
Explain the structure of carbonyl functional group
The carbonyl carbon atom:
In Clemmensen Reduction carbonyl compound is treated with:
Treatment of compound \[\begin{array}{cc}
\phantom{.....}\ce{O}\\
\phantom{.....}||\\
\ce{Ph - O - C - Ph}
\end{array}\] with \[\ce{NaOH}\] solution yields
(i) Phenol
(ii) Sodium phenoxide
(iii) Sodium benzoate
(iv) Benzophenone
Arrange the following in decreasing order of their acidic strength. Give explanation for the arrangement.
\[\ce{C6H5COOH, FCH2COOH, NO2CH2COOH}\]
Assertion: Compounds containing \[\ce{-CHO}\] group are easily oxidised to corresponding carboxylic acids.
Reason: Carboxylic acids can be reduced to alcohols by treatment with \[\ce{LiAlH4}\].
Among the given compound, the most susceptible to nucleophile attack at the at the carbonyl group
Which of the following is INCORRECT?
