Advertisements
Advertisements
प्रश्न
Write the reaction of aliphatic primary amine with nitrous acid.
उत्तर
Aliphatic primary amine react with nitrous acid (prepared in situ from NaNO2 and a mineral acid such as HCl) to form unstable aliphatic diazonium salts, which further produce alcohol and HCl with the evolution of N2 gas.
\[\ce{\underset{alcohol}{\underset{primary}{\underset{Aliphatic}{R - NH2}}} + \underset{acid}{\underset{Nitrous}{HNO2}} ->[NaNO2 + HCl] \underset{salt}{\underset{diazonium}{\underset{Aliphatic}{[R - \overset{+}{N2}\overset{-}{C}l]}}} ->[H2O] \underset{alcohol}{ROH} + HCl + N2 ^}\]
APPEARS IN
संबंधित प्रश्न
How do you convert the following: C6H5CONH2 to C6H5NH2
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Write reactions to prepare ethanamine from Acetonitrile.
Acetamide on reduction using Na/C2H5OH gives ____________.
The end product C of the following reaction is
\[\ce{C2H5NH2 ->[HNO2] A ->[PCl5] B ->[NH3][Alcohol] C}\]
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
Which of the following reactions is appropriate for converting benzamide to aniline?
Identify the INCORRECT statement regarding Hofmann bromamide reaction.
Quaternary ammonium salt is formed:
Amongst the following, the strongest base in aqueous medium is ______.
The best reagent for converting 2–phenylpropanamide into 2-phenylpropanamine is ______.
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
What is the best reagent to convert nitrile to primary amine?
Identify A and B in the following reaction.
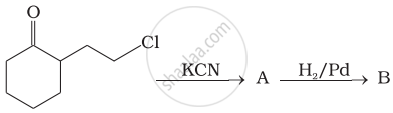
How will you bring out the following conversion?

Ethylamine can be prepared by the action of bromine and caustic potash on which compound?
Methyl amine on reaction with chloroform in the presence of NaOH gives ______.
Write a short note on the following:
Ammonolysis
Write the name of reduction product formed when ethyl cyanide is treated with sodium and alcohol.
Write a short note on Ammonolysis.
