Advertisements
Advertisements
प्रश्न
Write the reactions involved when D-glucose is treated with the following reagent:
(CH3CO)2O
उत्तर
\[\begin{array}{cc}
\phantom{...............}\ce{CHO}\phantom{...................}\ce{CHO}\phantom{....}\ce{O}\phantom{..}\\
\phantom{..............}|\phantom{.......................}|\phantom{........}||\\
\phantom{.....................}\ce{(CHOH)4->[Acetic anhydride](CH-O-C-CH3)4}\\
\phantom{......}|\phantom{.......................}|\\
\phantom{......................}\ce{\underset{\text{D-glucose}}{CH2OH}}\phantom{................}\ce{CH2-O-C-CH3}\\
\phantom{.................................................}||\\
\phantom{.................................................}\ce{\underset{\text{Glucose penta acetate}}{O}}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
How many moles of acetic anhydride will be required to form glucose pentaacetate from 2M of glucose?
(a) 2
(b) 5
(c) 10
(d) 2.5
Fill in the blanks by choosing the appropriate word/words from those given in the brackets:
(iodoform, acetaldehyde, positive, greater, acidic, acetone, disaccharide, negative, increases, glucose, decreases, chloroform, polysaccharide, lactose, lesser, basic, cationic hydrolysis, anionic hydrolysis)
Sucrose is a _________ and yields upon hydrolysis, a mixture of ________ and fructose.
Answer the following question.
What is the basic structural difference between glucose and fructose?
Write the reactions involved when D-glucose is treated with the following reagent:
H2N-OH
The following compound can be called as:
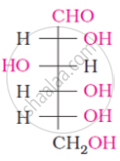
What is the most abundant organic compound on earth?
The symbols D and L represents ____________.
Which of the following properties of glucose cannot be explained by its open chain structure?
(i) Glucose does not form hydrogen sulphite with NaHSO3.
(ii) On oxidation with HNO3 glucose gives saccharic acid.
(iii) Glucose is found to exist in two different crystalline forms which are named as α and β.
The α-D glucose and β-D glucose differ from each other due to difference in carbon atom with respect to its ____________.
Assertion: D (+) – Glucose is dextrorotatory in nature.
Reason: ‘D’ represents its dextrorotatory nature.
