Advertisements
Advertisements
Question
40g of ice at 0°C is used to bring down the temperature of a certain mass of water at 60°C to 10°C. Find the mass of water used.
[Specific heat capacity of water = 4200 J kg-1 °C-1]
[Specific latent heat of fusion of ice = 336 × 103 J kg-1]
Solution
Let mass of water used = m gm.
By principal of calorimetry,
Heat given = Heat taken
m1c1ΔQ1 = mL + m2c2ΔQ2
m × 4.2 × (60 - 10) = 40 × 336 + 40 × 4.2 × (10 - 0)
m × 4.2 × 50 = 40 × 336 + 1680
m = 72 gm
APPEARS IN
RELATED QUESTIONS
How is the transference of heat energy by radiation prevented in a calorimeter?
Explain the melting point ?
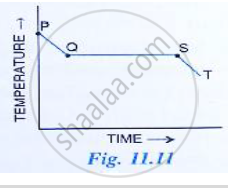
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
Some heat is given to 120 g of water and its temp. rises by 10 K. When the same amount of heat is given to 60 g of oil, its temp. rises by 40 K. The Sp. heat of water is 4200 J/kgK. Calculate:
(a) The amount of heat in joules given to water,
(b) The specific heat capacity of the oil.
40 g of ice at -16°C is dropped into water at 0°C, when 4 g of water freezes into ice. If specific heat capacity of ice is 2100 J/kg°C, what will be the latent heat of fusion of ice?
In an experiment. 17 g of ice is used to bring down the temp. of 40 g of water from 34°C to its freezing point. The sp. heat capacity of water is 4.25 J/g°C. Calculate sp. latent heat of ice.
Fill in the following blank using suitable word:
1 cal = .......... J
Why does the heat supplied to a substance during its change of state not cause any rise in its temperature?
Name the greenhouse gases.
A calorimeter is a device used to measure the ______.
