Advertisements
Advertisements
Question
A mass of 200 g of mercury at 100°C is poured into water at 20°C. If the final temperature of the mixture is 25°C, find the mass of water. (Sp. heat cap. of mercury is 140 J/kgK).
Solution
Let the mass of water be m.
Heat lost by mercury = Heat gained by water
0.2 x 140 x (100 - 25) = m x 4200 x (25 - 20)
or, m = `(0.2 xx 140 xx 75)/(4200 xx 5)` = 0.1 kg = 100 g
APPEARS IN
RELATED QUESTIONS
Name the law on which the principle of mixtures is based
Describe an experiment to show that there is absorption of heat energy when the ice melts.
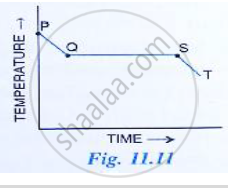
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
(i) Name the physical quantity which is measured in calories.
(ii) How is calorie related to the S.I. unit of that quantity ?
A bucket contains 10 liters of water at 80°C. Cold water at 25°C is run from a tap into the bucket of hot water for 20 seconds and the temp. Of the water in the bucket falls to 50°C.
(a) Calculate the rate at which cold water came out of the tap.
(b) State an assumption made in the above calculation.
Fill in the following blank using suitable word:
Whenever mechanical work is done, .......... Is produced.
Fill in the following blank using suitable word:
Steam at 100oC is hotter than water at ............
The process of converting a substance from solid state to gaseous state is called condensation.
