Advertisements
Advertisements
Question
A system goes from P to Q by two different paths in the P-V diagram as shown in figure. Heat given to the system in path 1 is 1000 J. The work done by the system along path 1 is more than path 2 by 100 J. What is the heat exchanged by the system in path 2?
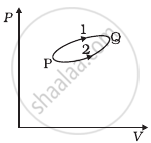
Solution
According to the first law of thermodynamics,
∆Q = AU + ∆W.
Let us apply this for each path.
For path 1: Heat given Q1 = + 1000 J
Let work done for path 1 = W1
For path 2: Work done (W2) = (W1 – 100) J
Heat has given Q2 = ?
A change in internal energy between two states for the different paths is the same.
∴ ∆U = Q1 – W1 = Q2 – W2
1000 – W1 = Q2 – (W1 – 100)
⇒ Q2 = 1000 – 100 = 900 J
APPEARS IN
RELATED QUESTIONS
The first law of thermodynamics is a statement of ____________ .
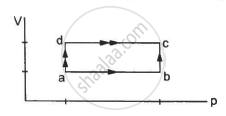
When a system is taken through the process abc shown in figure, 80 J of heat is absorbed by the system and 30 J of work is done by it. If the system does 10 J of work during the process adc, how much heat flows into it during the process?
An adiabatic vessel of total volume V is divided into two equal parts by a conducting separator. The separator is fixed in this position. The part on the left contains one mole of an ideal gas (U = 1.5 nRT) and the part on the right contains two moles of the same gas. Initially, the pressure on each side is p. The system is left for sufficient time so that a steady state is reached. Find (a) the work done by the gas in the left part during the process, (b) the temperature on the two sides in the beginning, (c) the final common temperature reached by the gases, (d) the heat given to the gas in the right part and (e) the increase in the internal energy of the gas in the left part.
Based on first law of thermodynamics which of the following is correct.
120 J of heat is added to a gaseous system, whose internal energy change is 60 J, then the amount of external work done is ____________.
Consider a cycle followed by an engine (Figure)
1 to 2 is isothermal
2 to 3 is adiabatic
3 to 1 is adiabatic
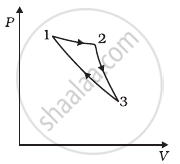
Such a process does not exist because ______.
- heat is completely converted to mechanical energy in such a process, which is not possible.
- mechanical energy is completely converted to heat in this process, which is not possible.
- curves representing two adiabatic processes don’t intersect.
- curves representing an adiabatic process and an isothermal process don’t intersect.
The initial state of a certain gas is (Pi, Vi, Ti). It undergoes expansion till its volume becomes Vf. Consider the following two cases:
- the expansion takes place at constant temperature.
- the expansion takes place at constant pressure.
Plot the P-V diagram for each case. In which of the two cases, is the work done by the gas more?
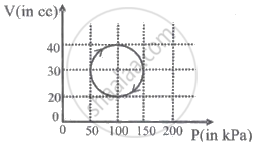
A system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is ______.
Mathematical equation of first law of thermodynamics for isochoric process is ______.
104 J of work is done on a certain volume of a gas. If the gas releases 125 kJ of heat, calculate the change in internal energy of the gas.
