Advertisements
Advertisements
Question
Accomplish the following conversion:
Benzamide to toluene
Solution
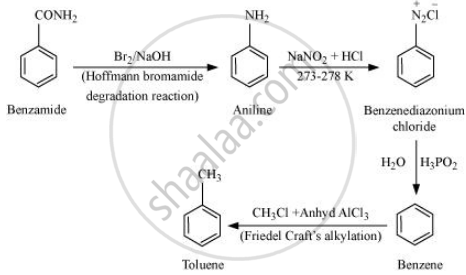
APPEARS IN
RELATED QUESTIONS
An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions :

Mendius reaction is used to convert _____________
Name the process of breaking C-X bond by ammonia in preparation of amines.
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives ______.
Best method for preparing primary amines from alkyl halides without changing the number of carbon atoms in the chain is ______.
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
How will you carry out the following conversion?
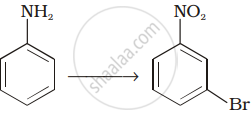
Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
Assertion: Hoffmann’s bromamide reaction is given by primary amines.
Reason: Primary amines are more basic than secondary amines.
Assertion: Aromatic 1° amines can be prepared by Gabriel Phthalimide Synthesis.
Reason: Aryl halides undergo nucleophilic substitution with anion formed by phthalimide.
Describe Gabriel's phthalimide synthesis. (Give reaction)
Ethylamine can be prepared by the action of bromine and caustic potash on which compound?
Reduction of nitro alkanes yields which compound?
In the given reaction what is the X?
\[\begin{array}{cc}
\ce{O}\phantom{.......................}\\
||\phantom{.......................}\\
\phantom{}\ce{R - C - OH <-[H3O] Χ ->[H] RCH2NH2}
\end{array}\]
Methyl amine on reaction with chloroform in the presence of NaOH gives ______.
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Write short note on the following:
Ammonolysis
