Advertisements
Advertisements
Question
Accomplish the following conversions: Benzyl chloride to 2-phenylethanamine
Solution

APPEARS IN
RELATED QUESTIONS
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Answer in one sentence.
Which amide does produce ethanamine by Hofmann bromamide degradation reaction?
Answer in one sentence.
Predict the product of the following reaction.
\[\ce{Nitrobenzene ->[Sn/conc.HCl]?}\]
Write the order of reactivity of alkyl halides with ammonia.
Write reactions to bring about the following conversions.
Acetamide to Ethylamine
Write reactions to bring about the following conversions.
Acetamide to methylamine
Acetamide on reduction using Na/C2H5OH gives ____________.
Alkyl cyanides on reduction by sodium and ethanol give primary amines. This reaction is called as ____________.
Identify the product obtained, when benzamide is treated with bromine and aqueous sodium hydroxide.
Which of the following amines forms a clear solution when treated with benzene sulphonyl chloride and excess of potassium hydroxide?
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Alkyl halides are insoluble in water.
Reason (R): Alkyl halides have halogen attached to sp3 hybrid carbon.
Select the most appropriate answer from the options given below:
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
Hoffmann Bromamide Degradation reaction is shown by ______.
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
Which of the following reactions are correct?
(i)
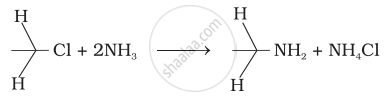
(ii)
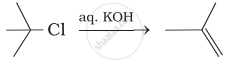
(iii)
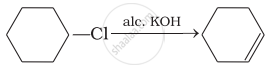
(iv)
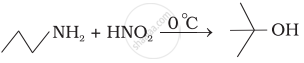
Assertion: Aromatic 1° amines can be prepared by Gabriel Phthalimide Synthesis.
Reason: Aryl halides undergo nucleophilic substitution with anion formed by phthalimide.
Describe Gabriel's phthalimide synthesis. (Give reaction)
Acetamide and ethyl amide can be distinguished by reacting with.
Which of the following reaction DOES NOT involve Hoffmann bromamide degradation?
