Advertisements
Advertisements
Question
Aluminium is extracted from its chief ore bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Name a chemicals used for dissolving aluminium oxide. In which state of sub-division is the chemical used?
Solution
Fluorspar and cryolite act as solvent. The percentage by weight composition is as follows:
Alumina - 20% by mass
Cryolite - 60% by mass
Fluorspar - 20% by mass
APPEARS IN
RELATED QUESTIONS
For the substance given below, describe the role played in the extraction of aluminium.
Sodium hydroxide
| Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction. |
Name a chemical used for dissolving aluminium oxide. In which state is the chemical used?
Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is
extracted from it by electrolytic reduction
Mention one reason for the use of aluminium in thermite welding ?
Name the following:
The substance added along with aluminium in the Hall-Heroult's process.
Give reason for the following:
Nitric acid can be stored in aluminium containers.
How is ore purified (give equations also)
How is aluminium obtained from pure ore?
The sketch below illustrates the refin ing of aluminium by Hoope's process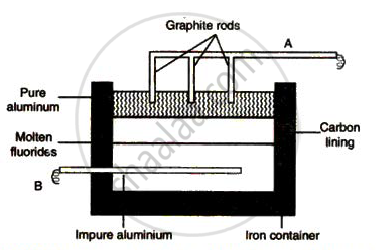
(a) Which of A and B is th e cathode and which one is the anode?
(b) What is the electroly te in the tank?
( c) What material is used for th e cathode?
Describe the role played in the extraction of aluminum:
Sodium hydroxide
Answer the following question:
Write the chemical formula of one main ore of iron and aluminium.
