Advertisements
Advertisements
Question
An alkene (A) on ozonolysis gives propanone and aldehyde (B). When (B) is oxidised (C) is obtained. (C) is treated with Br2/P gives (D) which on hydrolysis gives (E). When propanone is treated with HCN followed by hydrolysis gives (E). Identify A, B, C, D and E.
Solution
1. 2-methyl-but-2-ene (A) on ozonolysis gives propanone and acetaldehyde (B)
\[\begin{array}{cc}
\ce{CH3}\phantom{............................}\\
|\phantom{...............................}\\
\ce{\underset{\underset{(A)}{(2-methyl-but-2-ene)}}{CH3 - CH = C - CH3} ->[O3][Zn/H2O] \underset{\underset{(B)}{(Acetaldehyde)}}{CH3CHO} + CH3 - C - CH3}\\
\phantom{............................................}||\\
\phantom{............................................}\ce{\underset{(Propanone)}{O}}
\end{array}\]
2. Acetaldehyde (B) is oxidised to give acetic acid (C), which on further treated with Br2/P give monobromo acetyl bromide (D) which on hydrolysis gives monobromo acetic acid (E).
\[\begin{array}{cc}
\ce{\underset{\underset{(B)}{(Acetaldehyde)}}{CH3CHO} ->[H^+/K2Cr2O7][(O)] \underset{\underset{(C)}{(Acetic acid)}}{CH3COOH} ->[Br2/P][Excess] CH2COBr}\\
\phantom{.................................}|\\
\phantom{...................................}\ce{\underset{\underset{(D)}{(Monobromo acetyl bromide)}}{Br}}
\end{array}\]
3. Propanone is treated with HCN followed by hydrolysis to gives 2-methyl-2-hydorxy propanoic acid (E)
\[\begin{array}{cc}
\ce{COOH}\\
|\\
\ce{CH3 - C - CH3 ->[(i) HCN][(ii) H^+/H2O] CH3 - C - CH3}\\
\phantom{.}||\phantom{...........................}|\phantom{.}\\
\phantom{...........}\ce{\underset{(Propanone)}{O}}\phantom{............}\ce{\underset{\underset{(E)}{(2-methyl-2-hydorxy propanoic acid)}}{OH}}\phantom{}
\end{array}\]
| A | \[\begin{array}{cc} \ce{CH3 - CH = C - CH3}\\ \phantom{.......}|\\ \phantom{........}\ce{CH} \end{array}\] |
2-methyl-but-2-ene |
| B | CH3CHO | Acetaldehyde |
| C | CH3COOH | Acetic acid |
| D | Br – CH2 – COBr | Monobromo acetyl bromide |
| E | CH3C(OH) (COOH) CH3 | 2-methyl-2-hydorxy propanoic acid |
APPEARS IN
RELATED QUESTIONS
The IUPAC name of
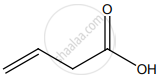
How is propanoic acid is prepared to start from alcohol?
How is propanoic acid is prepared to start from an alkyl halide?
How is propanoic acid is prepared to start from an alkene?
Oxidation of ketones involves carbon-carbon bond cleavage. Name the product(s) is/are formed on oxidising 2, 5-dimethyl hexan-3-one using a strong oxidising agent.
