Advertisements
Advertisements
Question
Answer the following in brief.
How instantaneous rate of reaction is determined?
Solution
i. To determine the instantaneous rate of a reaction the progress of a reaction is followed by measuring the concentrations of a reactant or product for different time intervals.
ii. The concentration of a reactant or a product is plotted against time.
iii. A tangent drawn to the curve at time t1 gives the rate of the reaction. The slope thus obtained gives the instantaneous rate of the reaction at time t1.
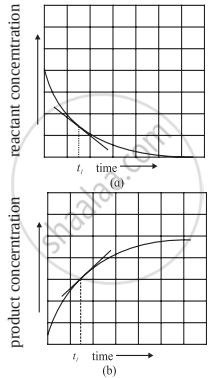
Determination of instantaneous rate
iv. Mathematically the instantaneous rate is expressed by replacing Δ by derivative dc/dt in the expression of the average rate.
Instantaneous rate = `("d"["C"])/"dt"`
v. For a reaction \[\ce{A → B}\]
Instantaneous rate of consumption of A at time t = `-("d"["A"])/"dt"`
Instantaneous rate of the formation of B at time t = `("d"["B"])/"dt"`
Therefore, instantaneous rate of the reaction at time t = `-("d"["A"])/"dt"` = `("d"["B"])/"dt"`
RELATED QUESTIONS
Choose the most correct option.
The rate law for the reaction aA + bB → P is rate = k[A] [B]. The rate of reaction doubles if _________.
Answer the following in one or two sentences.
For the reaction,
\[\ce{N2_{(g)} + 3H2_{(g)} -> 2NH3_{(g)}}\],
what is the relationship among `("d"["N"_2])/"dt"`, `("d"["H"_2])/"dt"` and `("d"["NH"_3])/"dt"`?
Rate constant for the reaction \[\ce{2N2O5 -> 4NO2 + O2}\] is 4.98 × 10–4 s–1. Find the order of reaction?
In a chemical reaction, \[\ce{2A + B -> C + D}\] Find the INCORRECT statement.
If concentration of reactant 'A' is increased by 10 times the rate of reaction becomes 100 times. What is the order of reaction, if rate law is, rate = k[A]x?
For the reaction \[\ce{H2_{(g)} + I2_{(g)} ⇌ 2HI_{(g)}}\], the average rate of reaction is expressed as ____________.
For the reaction \[\ce{A + 2B -> C}\], the instantaneous rate of reaction at a given instant can be represented by ____________.
In a reaction \[\ce{2A + P -> A2P}\], the reactant 'A' disappears at ____________.
The rate of disappearance of SO2 in the reaction \[\ce{2SO2 + O2 -> 2SO3}\] is 1.28 × 10−3 mol dm−3 s−1. Then the rate of formation of SO3 is ____________ mol dm−3 s−1.
For the reaction \[\ce{N2 + 3H2 ⇌ 2NH3}\]; if `(∆["NH"_3])/(∆"t")` = 2 × 10−4 mol dm−3 s−1, the value of `(-Δ["N"_2])/(∆"t")` would be ____________.
Which of the following expressions is CORRECT for the rate of formation of Br2?
\[\ce{5Br^-_{( aq)} + BrO^-_{3(aq)} + 6H^+_{( aq)} -> 3Br2_{(aq)} + 3H2O_{(l)}}\]
What is the average rate of reaction when the change in concentration of product is 0.05 Min 20 seconds?
For the reaction,
\[\ce{2NO(g) + O2(g) -> 2NO2(g) ->[d(NO2)][dt] = 0.052 mol dm^{-3} s^{-1}}\]
Calculate rate of consumption of NO (g).
What is the relation between `("d"["H"_2])/"dt" and ("d"["NH"_3])/"dt"`?
In the reaction A + 3B → 2C, the rate of formation of C is ______.
Define the rate of a reaction.
Explain the term instantaneous rate of a reaction.
For the reaction, \[\ce{3I^-(aq) + S2O^2-_8(aq) -> I^-_3(aq) + 2SO^2-_4(aq)}\]
`(d[SO_4^(2-)])/dt = 2.2 xx 10^(-2)` M/s at a particular time.
Calculate `(a) -(d[I^-])/dt (b) -(d[S_2O_8^(2-)])/dt (c) (d[I_3^-])/dt`
