Advertisements
Advertisements
Question
Answer the following question:
Which element of the first transition series has highest second ionisation enthalpy?
Solution
Out of all the elements of the first transition series copper has the highest second ionisation enthalpy.
Electronic configuration of Copper is: \[\ce{3d^10 4s^1}\]
After the Loss of first electron from the 4s copper acquires \[\ce{3d^10}\] configuration which is stable. Therefore, removal of second electron from the field 3-D orbital is very difficult and requires high amount of energy.
APPEARS IN
RELATED QUESTIONS
Out of Mn3+ and Cr3+, which is more paramagnetic and why ?
(Atomic nos. : Mn = 25, Cr = 24)
Explain briefly how +2 state becomes more and more stable in the first half of the first row transition elements with increasing atomic number?
Compare the stability of +2 oxidation state for the elements of the first transition series.
Explain why transition metals and their compounds act as a catalyst.
Out of \[\ce{Cu2Cl2}\] and \[\ce{CuCl2}\], which is more stable and why?
Assertion: Separation of \[\ce{Zr}\] and \[\ce{Hf}\] is difficult.
Reason: Because \[\ce{Zr}\] and \[\ce{Hf}\] lie in the same group of the periodic table.
Identify A to E and also explain the reactions involved.
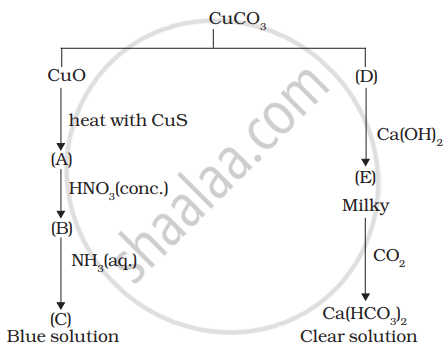
Answer the following question:
Which element of the first transition series has highest third ionisation enthalpy?
A complex in which dsp2 hybridisation takes place is
Assertion (A): Transition metals show their highest oxidation state with oxygen.
Reason (R): The ability of oxygen to form multiple bonds to metals.
