Advertisements
Advertisements
Questions
Answer the following.
Write a reaction to bring about the following conversions: Aniline into p-nitroaniline
Explain nitration of aniline. Write reactions for the preparation of p-nitroaniline.
Solution
Nitration: Direct nitration of aniline yields (p-nitroaniline) a mixture of ortho, meta, and para nitroanilines. In an acidic medium the -NH2 group is protonated to the -N+H3 group which is meta-directing and deactivating. Hence, a considerable amount of m-nitroaniline is obtained.

Preparation of p-nitroaniline: However, to get p-nitroaniline as a major product, the –NH2 group is first protected by acetylation and then nitration is carried followed by hydrolysis amide.

RELATED QUESTIONS
Answer the following
Write a reaction to convert Aniline into p-bromoaniline.
Answer the following
Complete the following reaction :
\[\ce{C6H5NH2 + Br2_{(aq)} -> ?}\]
Aniline on reaction with bromine water produces ________________
The following compound is a/an ____________.
Which of the following molecules form a Zwftter ion?
Identify the product 'B' in the following reaction.
\[\ce{Benzamide ->[Br2/KOH] A ->[Br2 water][298 K] B}\]
Which of the following molecule can form a zwitter ion?
Identify the CORRECT statements.
(I) Aniline reacts with bromine water at room temperature to give o-bromoaniline.
(II) Aniline undergoes Friedel Craft's reaction using aluminium chloride.
(III) Direct nitration of aniline yields, a mixture of ortho, meta and para nitro aniline.
(IV) Amino group is ortho and para directing and powerful ring activating group.
Identify the product obtained when chlorobenzene is heated with ammonia and Cu2O at 473 K under pressure.
Which of the following groups increases the basic strength of substituted aniline?
The major product obtained when aniline undergoes sulphonation using cone. sulphuric acid at 453-473 K is:
Aniline on reaction with Bromine water at room temperature gives ____________.
When propanamide is treated with bromine and aqueous sodium hydroxide, the compound formed is ____________.
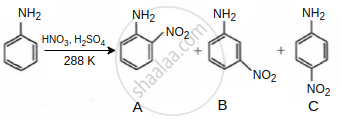
Consider the given reaction, percentage yield of:
In case of substituted aniline the group which decreases the basic strength is ______.
What is the action of aqueous bromine on aniline?
