Advertisements
Advertisements
Question
Arrange the following as per the instruction given in the brackets :
F, Cl, Br (Increasing electronegativity)
Solution
Br < Cl < F
APPEARS IN
RELATED QUESTIONS
Name the elements with highest and lowest electronegativity ?
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 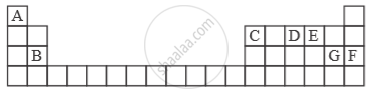
Which element forms electrovalent compound with G?
Fill in the blank
Most electronegative elements belong to ______ group.
Fill in the blank
The maximum electro negativity is shown by ______.
Fill in the blank
Reactivity ______ down the gruop for alkali metals with increase in electropositive character
Explain
Electronegativity of Cl is higher than S.
What property of an element is measured by electro negativity?
Arrange the following as per instruction given in the bracket.
Cs, Na, Li, K, Rb (decreasing electronegativity)
The element below sodium in the same group would be expected to have a ______ (lower/higher) electro-negativity than sodium and the element above chlorine would be expected to have a ______ (lower/higher) ionization potential than chlorine.
A group of elements in the periodic table is given below (Boron is the first member of the group and Thallium is the last). Answer the following question in relation to the given group of elements:
Which element would be expected to have the highest electronegativity?
