Advertisements
Advertisements
Question
Based on the group valency of elements state the formula lot the following giving justification for each :-
(i) Oxides of 1st group elements,
(ii) Halides of the elements of group 13, and
(iii) Compounds formed when an element of group 2 combines with, an element of group 16.
Solution
(i) Valency of group 1 elements: 1
Valency of oxygen: 2
Oxides of group 1 elements:
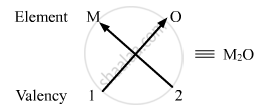
Formula of the oxides of group 1 is M2O, where M is the group 1 element and O is oxygen.
(ii) Valency of group 13 elements: 3
Valency of halogens: 1
Halides of group 13 elements:
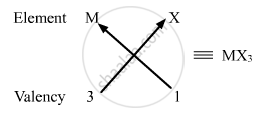
Formula of the halides of group 13 is MX3, where M is the group 13 element and X is halogen.
(iii) Valency of group 2 elements: 2
Valency of group 16 elements: 2
Compounds of group 2 and group 16 elements:
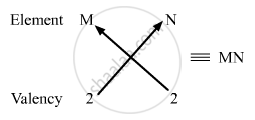
Formula of the compounds of group 2 and 16 is MN, where M is the group 2 element and N is the group 16 element.
APPEARS IN
RELATED QUESTIONS
The elements Be, Mg and Ca each having two electrons in their outermost shells are in periods 2, 3, and 4 respectively of the modern periodic table. Answer the following questions, giving justification in each case:-
(i) Write the group to which these elements belong.
(ii) Name the least reactive element.
(iii) Name the element having largest atomic radius.
Choose the most appropriate answer of the following:
Which of the following would weigh the least?
(A) 2-gram atoms of Nitrogen.
(B) 1mole of Silver
(C) 22.4 liters of oxygen gas at 1 atmospheric pressure and 273K
(D) 6.02 x 1023 atoms of carbon.
[Atomic masses: Ag=108, N=14, O=16, C=12]
How does the number of:
valence electrons and
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 
The ion of which element will migrate towards the cathode during electrolysis?
Explain
Size of atom changes when it loses or gain electron
Select the correct answer
Most reactive character among the elements given below is found in
Name and state the following with reference to the elements of the first three periods of the periodic table.
Valency of elements in Group 14 and 15.
Going across a period left to right, atomic size _____.
Name the elements in the correct order of their increasing atomic numbers present in the first, second, and third short periods of the periodic table.
State which of the elements are –
- metallic
- non-metallic
- noble gases
in each of the periods 2 and 3.
Name or state following with reference to the element of the first three periods of the periodic table.
A non-metal in period 2 having electronic configuration 2, 6.
