Advertisements
Advertisements
Question
Based on the group valency of element write the molecular formula of the following compound giving justification:
Halide of the elements of group thirteen
Solution
Halides of group 13 elements have a common formula of MX3, where M-metal and X- halogen element.
Example- AICI3, BF3. This is because the valency of group 13 elements is 3 and that of halogens is 1 so, to satisfy the combining capacity of aluminum or other group 13th elements three of halogens are required in the molecular formula.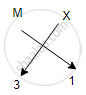
APPEARS IN
RELATED QUESTIONS
Define the term : Atomic size
Which of the following is the valence shell for the elements of second period of the modern periodic table?
(a) M shell
(b) K shell
(c) L shell
(d) N shell
How do the following change on moving from left to right in a period of the periodic table?
Give examples in support of your answer.
Chemical reactivity of elements ?
Complete the following sentences
The similarities in the properties of a group of elements are because they have the same …………………
How do you calculate the possible valency of an element from the electronic configuration of its atoms?
'In a group, the atomic radii increase with increasing period number', explain this statement and justify it with reference to group 17.
Name and state the following with reference to the elements of the first three periods of the periodic table.
Alkaline earth metal in period 3.
An element A has 2 electrons in its fourth shell. State:
is it a metal or non-metal
An element A has 2 electrons in its fourth shell. State:
is it an oxidising or reducing agent
Name the period which is the shortest period of the periodic table.
